- Search
| Ann Rehabil Med > Volume 48(2); 2024 > Article |
|
Abstract
Objective
Methods
Results
Conclusion
CONFLICTS OF INTEREST
The funding for this research was provided by M3 SOLUTIONS, CO., LTD, with Seoul National University Hospital serving as the lead research institution. This work falls under the category of commissioned research and the M3 Company played no role in the research design, data collection, analysis, or interpretation of results. The funding covered research expenses, equipment acquisition, and publication costs, with no influence on the research findings. The research was conducted independently, and the researchers maintained academic independence regarding the interpretation of the research results.
FUNDING INFORMATION
The funding for this research was provided by M3 SOLUTIONS, CO., LTD, with Seoul National University Hospital serving as the lead research institution (grant number: 0620214740). This work falls under the category of commissioned research.
AUTHOR CONTRIBUTION
Conceptualization: Lee GJ. Data curation: Kim H, Lee S. Methodology: Lee GJ. Formal analysis: Lee H. Funding acquisition: Lee GJ. Investigation: Lee H, Kim H. Project administration: Lee GJ. Resources: Lee GJ. Supervision: Lee GJ. Validation: Lee H. Visualization: Lee H. Writing – original draft: Lee H. Writing – review and editing: Lee H.
Fig. 1.
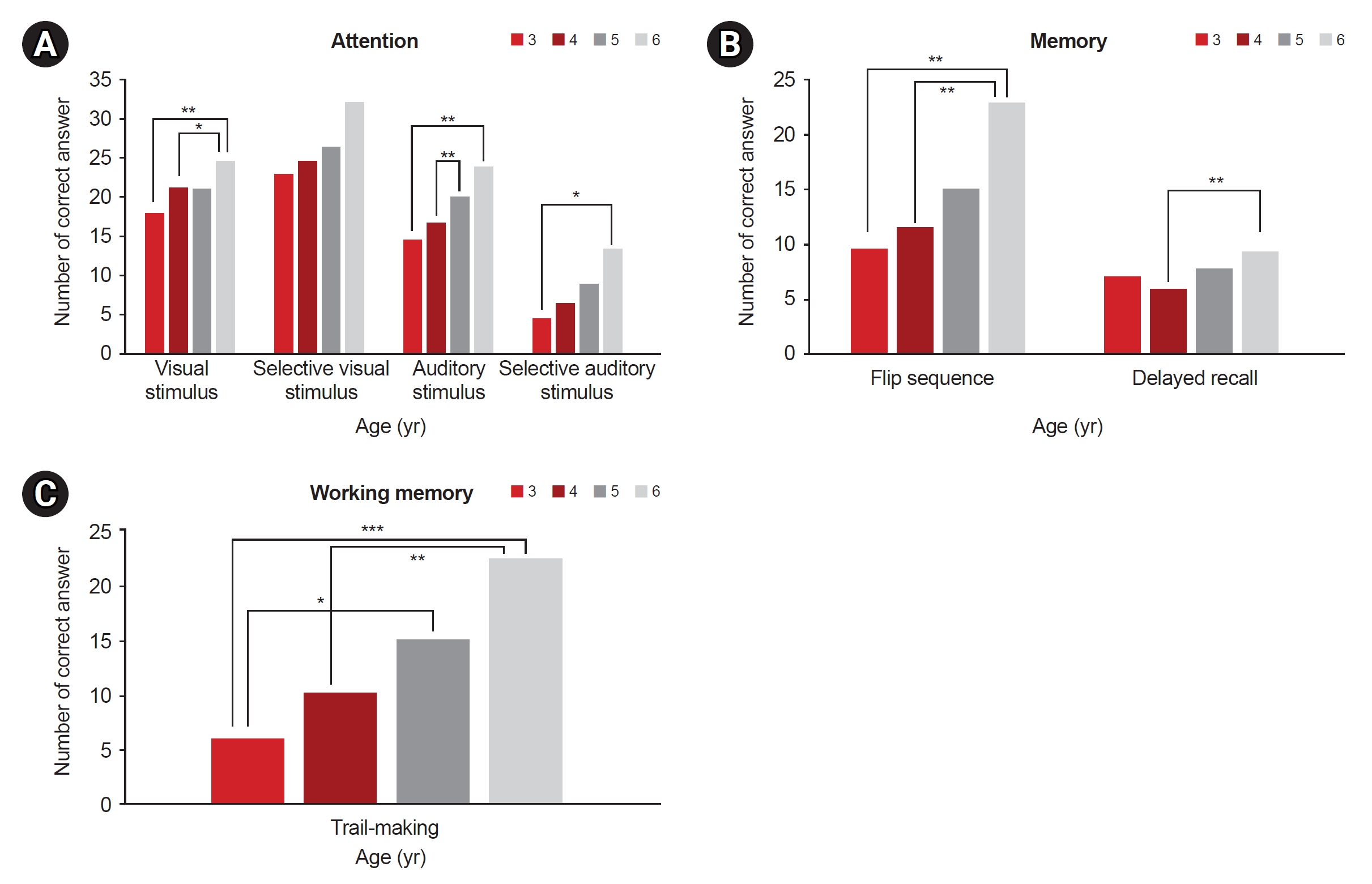
Fig. 2.
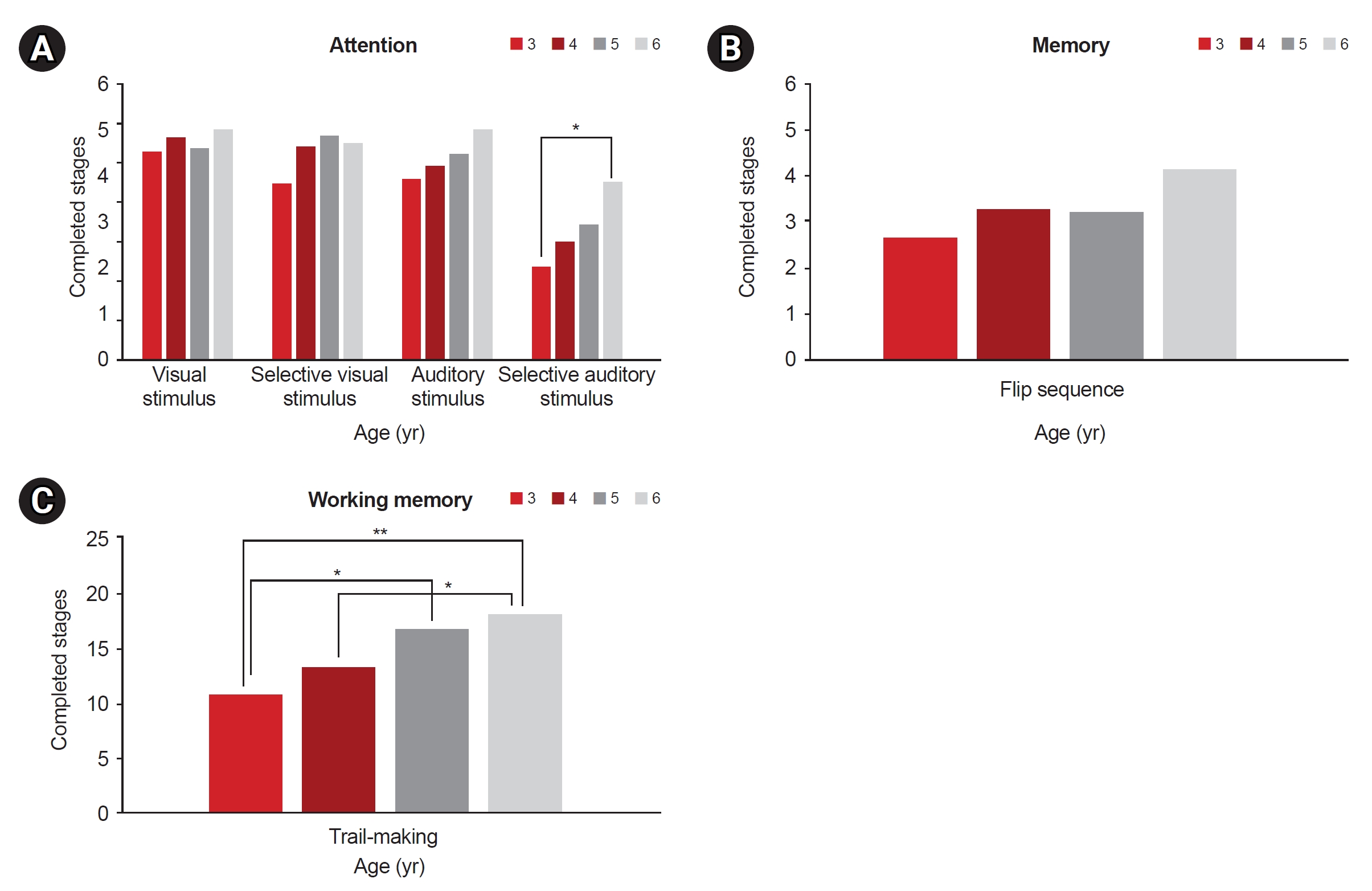
Table 1.
Table 2.
Table 3.
SD, standard deviation.
a)Visual stimulus test between age 3 and age 6, p=0.001.
b)Visual stimulus test between age 4 and age 6, p=0.011.
c)Auditory stimulus test between age 3 and age 6, p=0.001.
d)Auditory stimulus test between age 4 and age 6, p=0.001.
e)Selective auditory stimulus test between age 3 and age 6, p=0.010.
f)Flip sequence test between age 3 and age 6, p=0.001.
g)Flip sequence test between age 4 and age 6, p=0.001.
h)Delayed recall test between age 4 and age 6, p=0.001.
i)Trail making test between age 3 and age 6, p<0.001.
j)Trail making test between age 4 and age 6, p=0.002.
k)Trail making test between age 3 and age 5, p=0.017.