- Search
| Ann Rehabil Med > Volume 46(4); 2022 > Article |
|
Abstract
Objective
To analyze the effects of lymphovenous anastomosis (LVA) surgery after 1 year using the elastic index (EI) and volume.
Methods
This study was a retrospective study of 41 patients, with lymphedema, who underwent LVA surgery between July 2018 and June 2020. Limb circumference, used to determine the volume of the limb with lymphedema, and EI, which reflects tissue stiffness and measured using ultrasonography were measured for each patient before and 1 year after LVA surgery. To examine the effect of LVA, differences in the preoperative and 1-year postoperative volumes and EIs were analyzed using the Wilcoxon signed-rank test.
Results
The mean volume and EI of the dominant site in upper-extremity lymphedema were 2,309.4 cm3 and 1.4, respectively, preoperatively and 2,237.1 cm3 and 0.9, respectively, at 1 year postoperatively. The mean volume and EI difference of the dominant site 1 year after surgery was −16.6 cm3 (p=0.22) and −0.5 (p<0.001). The mean volume and EI of dominant site in lower-extremity lymphedema were 6,137.0 cm3 and 1.2, respectively, preoperatively, and 5,832.6 cm3 and 1.1, respectively, at 1 year postoperatively. The mean volume and EI difference of the dominant site 1 year postoperatively were −320.9 cm3 (p=0.04) and −0.2 (p=0.09), respectively.
Lymphedema treatment includes non-surgical methods, such as complete decongestion, and surgical methods. Recently, physiological surgical methods for lymphedema have emerged, one of which is lymphovenous anastomosis (LVA) surgery [1,2].
LVA surgery was first introduced in the 1960s for the treatment of lower extremity lymphedema. LVA is a surgical technique that improves lymphatic flow, such that the lymphatic vessels at the lymphedema site are connected to the surrounding venous blood vessels so that the lymph can be drained into the veins without being retained. In its early days, LVA surgery was limited to large blood vessels due to the primitive surgical equipment and methods available then. Moreover, pressure control posed a challenge. However, recent advancements in microsurgical techniques and medical equipment have made it possible to operate at a pressure of 0 mmHg using veins and lymphatic vessels with diameters of less than 1 mm [3,4].
As lymphedema progresses, the pressure of the subcutaneous tissue increases owing to increased fluid collection, leading to fibrotic changes and increased tissue stiffness. Therefore, the pressure increases, which consequently increases the volume, to eventually decrease the pressure and stiffness of the subcutaneous tissue. If lymphedema pressure is not properly controlled, this vicious cycle is repeated [5,6].
In this study, we used the elastic index (EI), which reflects tissue stiffness, to investigate the change in subcutaneous tissue pressure in the lymphedema region after surgery.
While many previous studies have demonstrated the effect of LVA surgery by changing the circumference and volume of lymphedema [7], this study is the first to reveal the effect of LVA surgery in terms of lymphedema pressure before and after LVA surgery.
We conducted a retrospective study of 41 patients, with upper- or lower-extremity lymphedema, who underwent LVA surgery between July 2018 and June 2020. The study was retrospectively carried out after the protocol approval from the Institutional Review Board of Ewha Womans University Mokdong Hospital (IRB No. 2021-09-005). Written informed consents were obtained.
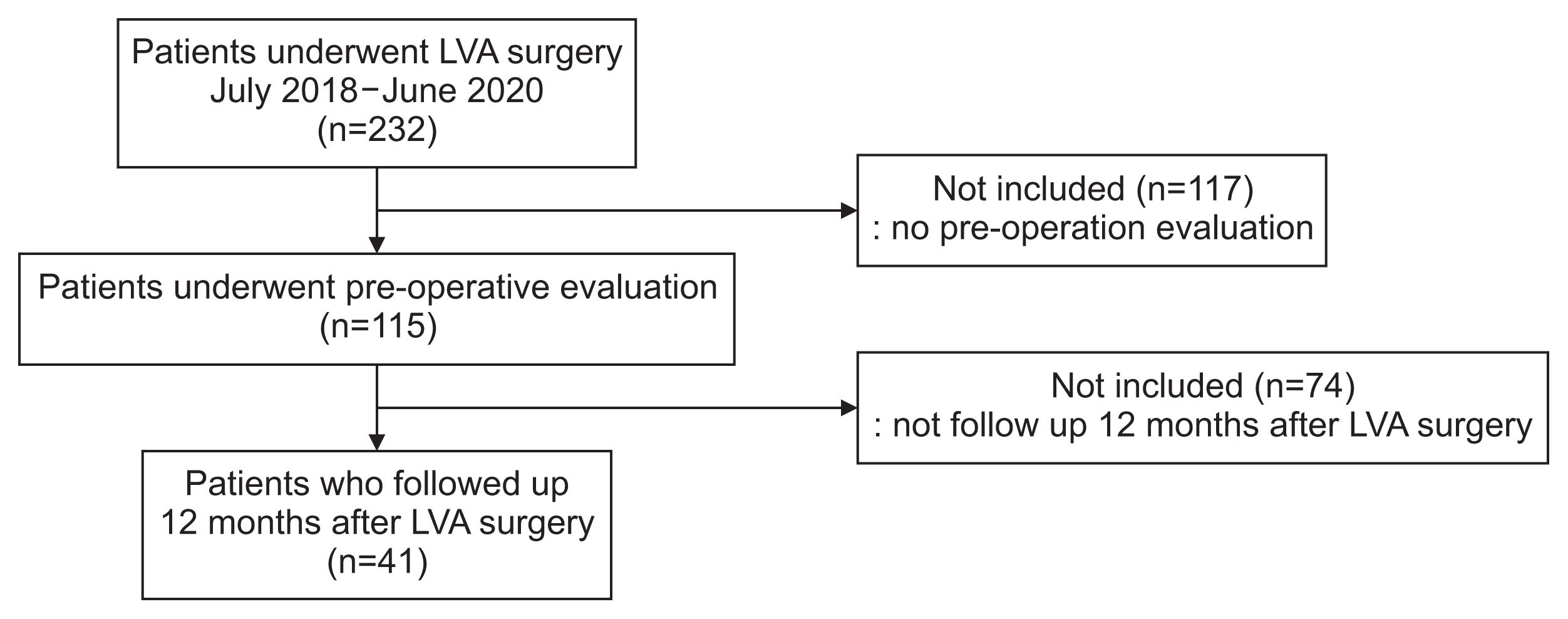
A total of 115 patients underwent preoperative LVA evaluation and were included in this study only if EI was evaluated using ultrasonography and the circumference of the upper or lower extremities with lymphedema before and 1 year after LVA surgery (Fig. 1).
Inclusion criteria for participants are (1) patients diagnosed with lymphedema, (2) patients with lymphatic vessels visible on indocyanine green (ICG) lymphangiography before LVA surgery, (3) patients who underwent LVA surgery, and (4) patients who were evaluated for circumference and EI using ultrasonography before and 1 year after LVA surgery. Exclusion criteria for participants are (1) patients who have not undergone LVA surgery, (2) patients who did not undergo preoperative evaluation (circumference and EI using ultrasonography), and (3) patients without follow-up 1 year after LVA surgery.
The following patient characteristics were obtained from medical records: age, sex, etiology of lymphedema, lymphedema duration, International Society of Lymphology (ISL) stage, indocyanine green dye (ICG) stage, number of LVA sites, laterality, number of LVA sites, body mass index (BMI), and episodes of lymphangitis before LVA surgery.
Limb circumference was measured for each patient before and 1 year after LVA surgery. Upper extremity circumference measurements were taken at 5 cm and 10 cm below the elbow crease and 5 cm and 10 cm above the elbow crease. Measurements of lower extremity circumference were taken 5 cm and 10 cm below the inferior margin of the patella and 5 cm and 10 cm above the superior margin of the patella.
Limb circumference was then used to determine the volume of the limb with lymphedema. We used this formula to obtain the volume of the dominant site by selecting thicker and more symptomatic limbs with lymphedema.
where h is 5 cm, r is 5 cm above or below the baseline (elbow crease or patellar margin) of the lymphedema-dominant site, and R is 10 cm above or below the baseline (elbow crease or patellar margin) of the lymphedema-dominant site).
In patients with upper extremity lymphedema, the dominant site was above the elbow crease in 9 patients (42.9%) and below the elbow crease in 12 patients (57.4%). In patients with lower extremity lymphedema, the dominant site was above the superior margin of the patella in 15 patients (75%) and below the inferior margin of the patella in five patients (25%).
Ultrasound elastography is a reliable tool for assessing the relative or absolute stiffness of soft tissues as an objective method for evaluating their elasticity. As lymphedema progresses, dermal fibrotic changes occur, which increase the pressure and stiffness of the soft tissues. Therefore, the EI for lymphedema is expected to reflect the stiffness of the lymphedema. It can also indirectly estimate the fluid accumulation of lymphedema by modifying the elasticity of the subcutaneous tissue; therefore, it is used to evaluate the status of lymphedema [8,9]. In addition, since ultrasound results are quite consistent with the clinical course, it may help objectively evaluate the course of treatment [10].
One method used in ultrasound elastography includes strain imaging (SI) or shear wave elastography imaging (SWEI) [11,12]. The EI was measured using the compression technique among the SIs using LOGIQ S7 Expert (GE Healthcare, Seoul, South Korea) [13], and it was measured for each patient 1 year before and after LVA surgery. It was performed by two examiners skilled in measuring EI using ultrasonography. The EI was measured 10 cm below the elbow crease and 10 cm above the elbow crease in the upper extremity lymphedema-dominant site. In the lower-extremity lymphedema-dominant site, EI was measured 10 cm below the inferior margin of the patella and 10 cm above the superior margin of the patella.
To examine the effect of LVA, differences in the preoperative and 1-year postoperative volumes and EIs were analyzed using the Wilcoxon signed-rank test. The results were analyzed using IBM SPSS (version 22; IBM Corp., Armonk, NY, USA). Statistical significance was set at p<0.05.
Of 41 patients, 21 (51.2%) had upper-extremity lymphedema and 20 (49.8%) had lower-extremity lymphedema. All patients with upper-extremity lymphedema were women, and 19 patients (95%) with lower-extremity lymphedema were women (Table 1). The mean age was 52.2 years.
The majority, 21 patients (51.2%), reported a history of breast cancer, 8 (40%) were treated for cervical cancer, 5 (25%) were treated for uterine cancer, and 4 (20%) had endometrial cancer (Table 1).
In upper-extremity lymphedema, the preoperative mean volume of dominant site was 2,309.4±620.6 cm3 and the postoperative mean volume of dominant site was 2,237.1±658.1 cm3 at 1 year after surgery. The mean volume difference of the dominant site 1 year after surgery was −16.6 cm3 (−224.8 to 69.4 cm3) (p=0.22) (Table 2).
And in lower-extremity lymphedema, the preoperative mean volume of dominant site was 6,137.0±2,121.4 cm3 and the postoperative mean volume of dominant site was 5,832.6±2,028.9 cm3 1 year after surgery. The mean volume difference of the dominant site 1 year after surgery was −320.9 cm3 (−744.8 to 8.5 cm3) (p=0.04) (Table 2).
When comparing the volume difference of the dominant site before and 1 year after LVA surgery, 13 patients (61.9%) with upper-extremity lymphedema and 15 patients (75%) with lower-extremity lymphedema showed a decrease in volume 1 year after surgery. However, compared with before surgery, the volume increased in 8 patients (38.1%) with upper-extremity lymphedema and in 5 patients (25.0%) with lower-extremity lymphedema (Table 3).
In upper-extremity lymphedema, the preoperative mean EI of dominant site was 1.4±0.5, and the postoperative mean EI of dominant site was 0.9±0.3, 1 year after surgery. The mean EI difference of the dominant site at 1 year after surgery was −0.5 (−0.8 to 0.3) (p<0.001) (Table 4).
In lower-extremity lymphedema, the preoperative mean EI of the dominant site was 1.2±0.3, and the postoperative mean EI of the dominant site was 1.1±0.2, 1 year after surgery. The mean EI difference of the dominant site at 1 year after surgery was −0.2 (−0.4 to 0.2) (p=0.09) (Table 4).
When comparing the EI difference of the dominant site before and 1 year after LVA surgery, 19 patients (90.4%) with upper-extremity lymphedema and 12 patients (60%) with lower-extremity lymphedema showed a decrease in EI 1 year after surgery. Compared with preoperative values, there was no change in EI in 1 patient (4.8%) with upper extremity lymphedema and 2 patients (10%) with lower extremity lymphedema. In addition, compared with before surgery, the EI increased in 1 patient (4.8%) with upper extremity lymphedema and in 6 patients (30%) with lower extremity lymphedema (Table 3).
Lymphedema is a chronic disease that requires continuous management and treatment after lymphedema develops. Conservative approaches, including complex decongestive therapy, are the primary treatment options for lymphedema. However, conservative approaches are often characterized by long treatment duration and the therapeutic effect largely depends on patient compliance. In addition to conservative treatments, two major surgical options are available for lymphedema treatment: the excisional surgical method and physiological surgical method. The excisional method includes liposuction and excisional debulking, followed by skin grafting. Meanwhile, the physiological surgical technique includes LVA surgery to connect veins and lymphatic vessels and vascularized lymph node transfer (VLNT) to the lesion. Recently, physiological surgical method has been spotlighted as a new treatment method.
Accordingly, this study aimed to evaluate whether LVA surgery, one of the physiological surgical methods, actually prevented the progression of lymphedema in patients with lymphedema, and this was confirmed through the volume and EI change values of lymphedema before and after surgery.
The change in volume before and 1 year after LVA surgery suggests that the mean volume of both upper- and lower-extremity lymphedema decreased when comparing the volume before and 1 year after LVA surgery at the dominant site of lymphedema [14].
When comparing the EI before and after LVA surgery at the dominant site of lymphedema, the mean EI value decreased for both upper- and lower-extremity lymphedema. Comparing the change of EI before and after LVA surgery, it can be seen that the rate of decreased or maintained EI in the upper-extremity lymphedema is higher than the lower-extremity lymphedema. Unlike the volume change, the EI change after LVA surgery had a statistically significant difference in upper-extremity lymphedema and showed a borderline significance level in lower-extremity lymphedema. Accurate postoperative measurement of volume difference is considered difficult because of the total volume of lower-extremity lymphedema being relatively larger than that of upper-extremity lymphedema. Consequently, although the tendency to reduce the pressure in lower-extremity lymphedema can be confirmed clinically, it remains difficult to prove statistically. Therefore, further research on methods that can measure the pressure change more accurately is needed.
This suggests that LVA surgery is more effective in reducing pressure than in reducing the volume of the lymphedema-dominant site. Previous studies have focused on volume or circumference reduction when analyzing the effects of LVA surgery. However, this study was the first to analyze the effect of LVA surgery on changes in pressure after LVA surgery.
As lymphedema progresses, fluid accumulates in the interstitial space, increasing the pressure and elasticity of lymphedema. If fluid accumulation continues due to impairment of lymph circulation, the circumference and volume of lymphedema increases due to increased pressure. Consequently, the pressure and elasticity decrease again, and patients think their symptoms improve, but the continuous accumulation of fluid causes lymphedema to increase in pressure and elasticity again, repeating a vicious cycle [5,6]. However, when lymph circulation is improved and fluid accumulation is reduced through LVA surgery, the increase in pressure and elasticity that occurs before LVA surgery is reduced, along with the volume.
When determining the effects of LVA surgery, changes in the circumference and volume of lymphedema are used as key factors. However, since lymphedema volume control is secondary to pressure control, pressure control should be the critical factor when determining the effect of LVA surgery in the future.
The study had some limitations. First, the sample size was small. In this study, the EI change 1 year after LVA surgery showed borderline statistical significance in lower-extremity lymphedema, so it is expected to show a statistically significant difference upon re-analysis after increasing the sample size.
Second, this study was conducted on patients 1 year after LVA surgery; however, it is necessary to analyze the trend of changes in volume and EI through long-term follow-up.
Third, EI measurement using ultrasound may cause errors, depending on the method used. Although the EI was measured by skilled examiners, the compression technique among the SIs used in this study may be more affected by the magnitude of the applied force and the skill of the examiner than the SWEI method [15]. Therefore, in future studies, it is necessary to analyze the effect of LVA surgery through EI measurements using the SWEI method.
In conclusion, LVA surgery tends to reduce the pressure and volume of a lymphedema-dominant site after surgery. To monitor lymphedema progression, focusing more on pressure and stiffness than on volume is necessary. Thus, LVA surgery may help prevent the progression of lymphedema.
AUTHOR CONTRIBUTION
Conceptualization: Lee JY, Bae HS. Methodology: Lee JY, Bae HS. Formal analysis: Lee JY. Funding acquisition: Lee JY, Bae HS. Project administration: Lee JY, Bae HS. Visualization: Lee JY, Kim SJ. Writing – original draft: Lee JY, Kim SJ. Writing – review and editing: Lee JY, Kim SJ, Woo KJ, Bae HS. Approval of final manuscript: all authors.
Table 1
Comparison of characteristics of patient
Table 2
Mean volume of lymphedema dominant site
| n | Volume (cm3) | p-value | |||
|---|---|---|---|---|---|
| Preoperative | 1-year postoperative | Δvolume | |||
| Upper lymphedemaa) | 21 | 2,309.4±620.6 | 2,237.1±658.1 | −16.6 (−224.8 to 69.4) | 0.28 |
| Lower lymphedemaa) | 20 | 6,137.0±2,121.4 | 5,832.6±2,028.9 | −320.9 (−744.8 to 8.5) | 0.04* |
Table 3
Difference of volume and the elastic index (EI) of lymphedema dominant site
| Upper lymphedema (n=21) | Lower lymphedema (n=20) | |
|---|---|---|
| Δvolume<0 | 13 (61.9) | 8 (38.1) |
| Δvolume>0 | 15 (75.0) | 5 (25.0) |
| ΔEI<0 | 19 (90.4) | 12 (60.0) |
| ΔEI>0 | 1 (4.8) | 6 (30.0) |
| ΔEI=0 | 1 (4.8) | 2 (10.0) |
REFERENCES
1. Maclellan RA, Couto RA, Sullivan JE, Grant FD, Slavin SA, Greene AK. Management of primary and secondary lymphedema: analysis of 225 referrals to a center. Ann Plast Surg 2015;75:197-200.

2. Park KE, Allam O, Chandler L, Mozzafari MA, Ly C, Lu X, et al. Surgical management of lymphedema: a review of current literature. Gland Surg 2020;9:503-11.



3. Venkatramani H, Shanmugakrishnan RR, Kumaran MS, Sabapathy SR. Surgical debulking, lymphatico venous anastomosis, vascularised lymph node transfer in lower limb lymphoedema. Plast Aesthet Res 2020;7:19.

4. Forte AJ, Khan N, Huayllani MT, Boczar D, Saleem HY, Lu X, et al. Lymphaticovenous anastomosis for lower extremity lymphedema: a systematic review. Indian J Plast Surg 2020;53:17-24.



5. Azhar SH, Lim HY, Tan BK, Angeli V. The unresolved pathophysiology of lymphedema. Front Physiol 2020;11:137.



7. Yodrabum N, Khaogate K, Chaikangwan I, Aojanepong C. Lymphaticovenular anastomosis for patients with lymphedema of the upper extremity at Siriraj Hospital: a quantitative analysis study. J Med Assoc Thai 2021;104:620-8.

8. Forte AJ, Huayllani MT, Boczar D, Cinotto G, McLaughlin SA. Ultrasound elastography use in lower extremity lymphedema: a systematic review of the literature. Cureus 2019;11:e5578.



9. DeJong HM, Abbott S, Zelesco M, Kennedy BF, Ziman MR, Wood FM. The validity and reliability of using ultrasound elastography to measure cutaneous stiffness, a systematic review. Int J Burns Trauma 2017;7:124-41.


10. Suehiro K, Morikage N, Murakami M, Yamashita O, Samura M, Hamano K. Significance of ultrasound examination of skin and subcutaneous tissue in secondary lower extremity lymphedema. Ann Vasc Dis 2013;6:180-8.



11. Shiina T, Nightingale KR, Palmeri ML, Hall TJ, Bamber JC, Barr RG, et al. WFUMB guidelines and recommendations for clinical use of ultrasound elastography: part 1: basic principles and terminology. Ultrasound Med Biol 2015;41:1126-47.


12. Sigrist RM, Liau J, Kaffas AE, Chammas MC, Willmann JK. Ultrasound elastography: review of techniques and clinical applications. Theranostics 2017;7:1303-29.



13. Ophir J, Cespedes I, Ponnekanti H, Yazdi Y, Li X. Elastography: a quantitative method for imaging the elasticity of biological tissues. Ultrason Imaging 1991;13:111-34.



- TOOLS