- Search
| Ann Rehabil Med > Volume 43(6); 2019 > Article |
|
Abstract
Objective
To investigate the effects of physical therapy interventions using strengthening and stretching exercise programs on pain and temporospatial gait parameters in patients with plantar fasciitis (PF).
Methods
Eighty-four patients with PF participated in the study and were randomly assigned to the strengthening or stretching exercise groups. All patients received 8 physical therapy interventions two times per week in the first 4 weeks and performed daily strengthening or stretching exercises three times per day. After 4 weeks, they continued the assigned exercise programs every day for 8 weeks. Pain visual analogue scale (VAS) scores at the worst and in the morning and temporospatial gait parameters were evaluated at the baseline, intermediate of the intervention, end of the intervention, and the first and second month follow-up.
Results
There were significant effects of the time on the worst pain, morning pain, cadence, stride time, stride length, total double support, and gait speed, but there was no effect on step width. In addition, the main effect of the group and the interaction effects of the time and the group were not found in any parameters. For intra-group comparisons, there were significant differences in worst pain, morning pain, cadence, and stride time among the assessment times in both groups. For inter-group comparisons, there were no significant differences in all parameters.
Plantar fasciitis (PF) is a common foot disorder involving pain and gait problems [1,2]. The typical pain of PF is located at the medial tubercle of the calcaneus and normally occurs during the first few steps in the morning or after a prolonged non-weight bearing activity. Pain can be reduced after continuing more steps or performing light activities, but it repeatedly returns with prolonged weight-bearing activities [3,4]. Previous studies have reported that heel pain occurs in about 10% of the American population, and 80% of this group was diagnosed with PF [2,5]. In the US healthcare system, more than 2 million patients with PF annually receive assessments and management for their symptoms [5,6]. Diagnosis of PF can be made through the patient history, clinical symptoms, foot questionnaires, and objective assessments such as pain level, palpation, muscle tightness, joint range of motion, or muscle strength [2,7]. In addition, diagnostic imaging may be used to exclude other causes of pain such as heel spurs or tissue inflammation [7,8].
The main problem in PF is heel pain during weightbearing activities, especially walking, which is the most essential function for daily living [9]. Walking problems in patients may result in restrictions in function, working, social participation, and quality of life (QoL) [2]. Patients with PF who have painful episodes at the heel commonly avoid weight bearing on the symptomatic foot and are at risk for developing antalgic gait [3]. Patients with PF tend to walk more slowly than healthy individuals in order to avoid or reduce pain [10]. They show significant decreases in cadence, gait speed, stride length, and increases in stride time [11,12]. In addition, the adaptation mechanism of antalgic gait may present in individuals in the chronic phase or a non-painful episode of PF [3,10]. As previously mentioned, foot pain from PF can reduce foot function and walking capacity, thus leading to decreased general health-related QoL [9]. The lack of walking or weight-bearing activities could result in the loss of muscle strength and muscle flexibility, development of an inactive lifestyle, and increased body weight [13]. Therefore, PF treatment is necessary to preserve QoL and prevent other potential problems [13].
Several treatment techniques have been conducted for patients with PF [1,3,4,6]. Generally, these patients start with non-surgical or conservative treatment. If this treatment cannot improve the symptoms after 6–12 months, surgical treatment will be considered [3]. Conservative treatments for PF usually include rest, anti-inflammatory drugs, shoe inserts, shoe wear modification, stretching exercises, and physical therapy. Examples of physical therapy are massage, mobilization, therapeutic ultrasound, and taping. However, there is controversy about the treatment benefits and there is insufficient evidence about their efficacy [1,2,4,14]. Among several conservative treatments, only gastrocnemius muscle and plantar fascia stretching were shown to have moderate treatment effects; they are often used in the treatment programs for patients with PF [1,4]. In 2006, Digiovanni et al. [15] reported that specific stretches of the plantar fascia for 8 weeks had the long-term benefit of reducing pain and function limitations, with a high rate of patient satisfaction.
Even though PF has several risk factors, an abnormal biomechanical structure is considered to play an important role. Abnormalities include tightness in the Achilles tendon and plantar fascia and reduced foot and ankle muscle strength [13,16]. Patients with PF often also have myofascial restrictions and muscle stiffness or tightness in the gastro-soleus complex and plantar fascia [17,18]. In 2015, Sullivan et al. [19] identified musculoskeletal factors in patients with PF compared to normal healthy people, and they found weakness in various muscles such as ankle evertors (peroneus brevis and longus) and toe flexors (flexor hallucis longus and brevis, flexor digitorum longus and brevis) [19]. In addition, weakness of the tibialis posterior muscle was also reported in previous studies [4,20]. Weakness of the intrinsic and extrinsic muscles of the foot may result in the recurrent symptoms of PF due to improper functioning of the muscle and joint positioning during walking [4,13,19]. Rathleff et al. [21] in 2014 reported that daily heel-raising exercises have superior effectiveness at 3 months than plantar-specific stretching. However, these exercises showed no difference in foot function at 1, 2, 6, or 12 months after this intervention.
The aforementioned exercise programs have an impact on PF recovery because they increase muscle strength and reduce muscle tightness. There was a limited number of studies comparing the effect of strengthening and stretching exercises; hence, it is still uncertain which exercises will improve more PF symptoms. Therefore, this study aimed to investigate the effects of strengthening and stretching exercise programs combined with a physical therapy intervention on pain intensity and temporospatial gait parameters in patients with PF. The hypothesis of this study was that the strengthening and stretching exercise programs may have different effects on reducing pain and improving gait performance.
A double-blind, randomized controlled trial was used in the study. The study was performed from July 2017 to February 2019 at the Physical Therapy Center, Faculty of Physical Therapy, Mahidol University, Thailand. The study protocol was approved by the Institutional Review Board of Mahidol University (No. MU-CIRB 2016/182.0211). Before participating in the study, the participants received explanations about the procedures and signed consent forms.
Patients with unilateral or bilateral PF were screened and recruited through printed and digital media. Participants were recruited if they met the following criteria: heel pain for more than 1 month; history of heel pain during the first few steps in the morning or after prolonged non-weight bearing activities [1,22]; pain with tenderness on palpation at the medial tubercle of calcaneus; and thickness of the plantar fascia greater than 4.0 mm, assessed by a portable digital ultrasound diagnosis imaging system (D-6600; Mindray Bio-Medical Electronics Co. Ltd., Zhenzhen, China) [22,23]. Participants were excluded if they had a history of back or lower limb surgery, fracture or trauma within 6 months, leg length discrepancy greater than 1 cm, corticosteroid injections within 6 months, pregnancy, and inability to perform the exercise program. In addition, participants were excluded if they were receiving any other intervention or performing other foot-specific exercises during the study. Furthermore, if the participants had Achilles tendinopathy, acute ankle sprains, tarsal tunnel syndrome, or patellofemoral syndrome during physical examinations, they were excluded. If palpation, pain in the center of calcaneus, and ultrasound indicated calcaneal spurs and heel pad syndrome, the participants were excluded. Patients with systematic diseases such as rheumatoid arthritis, fibromyalgia or neuropathy, neurological diseases, and diseases of the central or peripheral nervous system were excluded if they exhibited symmetrical pain and inflammation, general numbness, or a history of symptoms. These criteria were screened by a physical therapist with an experience of more than 5 years in the musculoskeletal field. A summary of the flowchart diagram is illustrated in Fig. 1.
Among the 102 eligible participants aged between 20 and 80 years, 18 were excluded according to the recruitment criteria due to Achilles tendinitis (n=3), acute ankle sprain (n=2), heel pad syndrome (n=4), pain in lower extremity (n=5), or inability to follow the research timeline (n=4). Therefore, 84 patients with PF participated in this study, and there were no dropouts. A stratified randomization table according to age (less or more than 50 years) and pain score (less or more than 5 out of 10 scores) was used to randomize the participants into the strengthening (n=42) or stretching (n=42) groups.
The researcher evaluated all participants five times, at the baseline, intermediate of the intervention, end of the intervention, and 1 and 2 month follow-ups after the end of the intervention. At the baseline, the demographic data collected by questionnaire included age, height, weight, PF characteristic, PF side, lower limb dominance, pain characteristic, and underlying diseases.
The participants were asked about their worst pain during the current day (worst pain) and their pain when taking the first few steps in the morning (morning pain) using the 10-cm visual analogue scale (VAS). The left and right ends of the line demonstrate ‘no pain’ as the ‘0 score’ and the ‘worst pain ever’ with the ‘10 scores’, respectively.
The temporospatial parameters of the patients were evaluated on a 3-m force distribution platform (The Zebris FDM-System-Gait Analysis; Zebris Medical GmbH, Isny, Germany) with a sampling frequency of 100 Hz and synchronized with a video camera (SC-1 SYNCCam; Zebris Medical GmbH). The video camera was placed at the end of the platform. The participants stood at the edge of the platform and were asked to walk barefoot at a comfortable speed to the other end of the platform. Data were collected for 3 trials, and the averaged data were used in the comparisons. The temporospatial gait parameters consisted of cadence (step/min), gait speed (m/s), stride time (s), stride length (m), step width (m), and double support time (%).
Both groups received the same conservative physical therapy intervention by the first physical therapist. This intervention consisted of therapeutic ultrasound and manual therapy eight times (two times per week over 4 weeks) followed by the assigned exercise program. The second and the third physical therapists instructed the participants to perform the strengthening and stretching exercises and reminded them to do the home-based exercise program three times per day. These two physical therapists only knew the details of their assigned exercise program; hence, they were blinded from the other exercise program. In addition, the participants also were blinded from the other exercise group. After the end of the intervention, participants continued their exercise programs at home for 2 months. All participants received illustrated instruction sheets with the details of the daily exercise program and its progression protocol. To monitor their compliance, we asked them to record their exercises in a logbook.
Therapeutic ultrasound (Sonopuls 492; Enraf-Nonius, Rotterdam, The Netherlands) with a 50% pulse mode at a frequency of 1.0 MHz and intensity of 1.5 W/cm2 was applied to the participants for 5 minutes with a slow circular movable technique on the most tender or painful area of the heel [24]. After finishing therapeutic ultrasound, manual therapy, plantar fascia stretching, and tissue mobilization [25], anteroposterior (AP) mobilization on the talocrural joint [25,26] and eversion-inversion mobilization on the subtalar joint [26] were performed.
The strengthening exercise program consisted of a toe curl exercise, ankle evertor exercise, ankle invertor exercise, and heel raise exercise [21,26]. The starting level of each exercise was selected individually by the physical therapist, evaluating participant performance. The exercise progression also based on the individual achievement of the previous level for each exercise. The toe curl exercise placed a kilogram sandbag on a towel for each additional level of progression. For the ankle evertor and invertor exercise, a harder TheraBand resistance band was placed at each level. The heel raise exercise progressed by its starting position; both feet with hand support, both feet without hand support, single foot with hand support, and single foot without hand support.
SPSS version 25 (IBM SPSS, Armonk, NY, USA) was used for statistical analysis. The statistical significance was estimated at p<0.05. The Kolmogorov-Smirnov goodness of fit test was used to test the data distribution; it showed a normal distribution.
The outcome measures were analyzed by the two-way repeated measure for ANOVA to investigate the main effects of time (at the baseline, intermediate of intervention, end of the intervention, first and second month follow-ups), main effects of the groups (strengthening and stretching), and interaction effects of time by group on the testing parameters. In addition, the Bonferroni posthoc analysis was used to find the pair of differences, and the independent sample t-test was used to find the differences between groups at each of the assessment times.
The sample size for this study was calculated based on the previous study [28]. The study investigated the effectiveness of low-level laser therapy to the improvement of gait speed in patients with PF (n=49) and showed that the average and standard deviation of gait speed in the experimental and control groups were 1.15±0.26 and 1.25±0.26 m/s, respectively. Using the G*Power program version 3.1.9.2 with the function of the t-test and setting the alpha error to 0.05 and power to 0.80, the estimated total sample number was 55 participants. Thus, the 84 samples that the present study collected were sufficient.
Demographic data of the participants in both groups are shown in Table 2. There were no significant differences (p>0.05) in age, gender, weight, height, worst pain, morning pain, PF side, PF characteristic, pain characteristic, and underlying disease between groups at the baseline.
There were significant effects of time on the worst pain (F(3.283, 269.201)=65.661, p<0.001), morning pain (F(2.812, 230.624)=87.421, p<0.001), cadence (F(3.066, 251.398)=7.663, p<0.001), stride length (F(2.986, 244.848)=9.239, p<0.001), stride time (F(2.872, 235.543)=8.687, p<0.001), total double support (F(3.607, 295.757)=3.601, p=0.009), and gait speed (F(2.862, 234.653)=14.411, p<0.001), but no significant effect of time on the step width (F(4, 298.537)=1.348, p=0.252). There were no significant effects on the group for worst pain (F(1, 82)=0.128, p=0.721), morning pain (F(1, 82)=0.276, p=0.601), cadence (F(1, 82)=0.088, p=0.768), step width (F(1, 82)=0.001, p=0.975), stride length (F(1, 82)=0.045, p=0.832), stride time (F(1, 82)=0.187, p=0.666), total double support (F(1, 82)=0.571, p=0.452), and gait speed (F(1, 82)=0.092, p=0.763). There were no significant interaction effects on the time and group for worst pain (F(3.283, 269.201)=0.313, p=0.833), morning pain (F(2.812, 230.624)=1.315, p=0.271), cadence (F(3.066, 251.398)=1.378, p=0.250), step width (F(4, 298.537)=0.611, p=0.655), stride length (F(2.986, 0.013)=1.924, p=0.127), stride time (F(2.872, 235.543)=1.407, p=0.242), total double support (F(3.607, 295.757)=0.851, p=0.484), and gait speed (F(2.862, 234.653)=1.325, p=0.268).
Table 3 shows intra- and inter-group comparisons for the pain intensity and the temporospatial gait parameters. Fig. 2 shows the changes in pain intensity, and Fig. 3 shows the changes in temporospatial gait parameters for both groups within 3 months. Tables 4 and 5 show pairwise comparisons of the pain intensity and the temporospatial gait parameters (except for the step width, which showed no difference) in the strengthening and stretching groups.
In the strengthening group, there were significant differences in the worst pain between the baseline and intermediate of the intervention (p<0.001), the baseline and end of the intervention (p<0.001), the baseline and first month follow-up (p<0.001), the baseline and second month follow-up (p<0.001), the intermediate of the intervention and first month follow-up (p=0.002), the intermediate intervention and second month follow-up (p<0.001), and the end of the intervention and second month follow-up (p=0.005). For morning pain, significant differences were found between the baseline and intermediate of the intervention (p<0.001), the baseline and end of the intervention (p<0.001), the baseline and first month follow-up (p<0.001), the baseline and second month follow-up (p<0.001), the intermediate and end of the intervention (p=0.028), the intermediate of the intervention and first month follow-up (p<0.001), the intermediate of the intervention and second month followup (p<0.001), the end of the intervention and second month follow-up (p<0.001), and then first and second month follow-ups (p=0.002). For cadence, a significant difference was found between the intermediate and end of the intervention (p=0.031). For stride time, significant differences were found between the intermediate of the intervention and first month follow-up (p=0.018), and the intermediate of the intervention and second month follow-up (p=0.020). In addition, no significant differences (p>0.05) of stride length, total double support, and gait speed among the times of assessment were found.
In the stretching group, there were significant differences in the worst pain baseline and intermediate of intervention (p=0.002), the baseline and end of the intervention (p<0.001), the baseline and first month followup (p<0.001), the baseline and second month followup (p<0.001), the intermediate of intervention and first month follow-up (p=0.004), the intermediate of intervention and second month follow-up (p<0.001), and the end of the intervention and second month followup (p=0.005). For morning pain, significant differences were found between the baseline and intermediate of the intervention (p=0.011), the baseline and end of the intervention (p<0.001), the baseline and first month followup (p<0.001), the baseline and second month follow-up (p<0.001), the intermediate and end of the intervention (p=0.001), the intermediate of the intervention and first month follow-up (p<0.001), the intermediate of the intervention and second month follow-up (p<0.001), the end of the intervention and first month follow-up (p=0.043), and the end of the intervention and second month follow-up (p=0.016). For cadence, significant differences were found between the baseline and first month followup (p=0.012) and between the baseline and second month follow-up (p=0.045). For stride length, significant differences were found between the baseline and intermediate of the intervention (p=0.047), the baseline and first month follow-up (p=0.002), and the intermediate of the intervention and second month follow-up (p=0.001). For stride time, significant differences were found between the baseline and first month follow-up (p=0.008) and between the intermediate of the intervention and second month follow-up (p=0.043). For double support time, a significant difference was found between the baseline and second month follow-up (p=0.030). For gait speed, significant differences were found between the baseline and intermediate of the intervention (p=0.010), the baseline and first month follow-up (p=0.001), the baseline and second month follow-up (p<0.001), and the end of intervention and second month follow-up (p=0.003).
In this study, patients with PF were randomly allocated to two groups to receive physical therapy treatment with the strengthening or stretching exercise programs for 12 weeks. The results showed that both groups attained similar enhancements in the reduction of pain and improvement of gait parameters. The worst pain and morning pain similarly decreased in both groups. This result was similar to the previous findings that showed the reduction of pain after patients with PF received stretching and strengthening exercises [27,29,30]. When compared to the baseline, the worst pain and morning pain started to show a significant reduction at the intermediate of the intervention and continued to reduce until the end of 3 months. Although the findings did not show a statistically significant difference between groups, the strengthening group had a slight decrease in morning pain in the first month, more than in the stretching group. The stretching group tended to have a slight increase in stride length at the 3-month follow-up, more than in the strengthening group. The effect of strengthening and stretching exercises on testing outcomes demonstrated in this study may suggest whether therapists select either strengthening or stretching exercise programs for recovering from pain and improving gait function in patients with PF. The selection may be decided based on the clinical characteristics of the patients, the availability of the instrument, and the proper location for each exercise protocol.
From our study, the change of morning pain in the first month of the strengthening group was 2.96 cm and that of the stretching was 2.29 cm. A previous study reported that the minimal clinically important difference (MCID) of the 10-cm VAS pain score was 3 cm [31] and the minimal important difference (MID) of 10-cm VAS pain was 0.8 cm for the average pain and 1.9 cm for the first step (or morning) pain [32], although the comparison between groups was not statistically different. But it may be implied that the level of morning pain was decreased with the clinical effects for the strengthening group more than that of the stretching group.
The present study found that both groups could improve their temporospatial parameters with no difference between the two exercise programs. The strengthening group showed improvements in cadence at the first month follow-up and in stride time at the second month follow-up. The stretching group showed improvements in cadence and stride time at the first month followup, improved double support time at the second month follow-up, and improved stride length and gait speed at the intermediate of the intervention. From the previous study, the recommended criteria for meaningful change in stride length was 0.005 m [33]. In our results, the changes of stride length in the strengthening and stretching were 0.02 and 0.05 m, respectively. This may identify the important changes over time for both exercise programs in the clinic. For the overall change in temporospatial parameters, both groups showed improvements in the same direction. We found that the restoration of gait parameters returned to the normal values [34]. At the end of the study, cadence, gait speed, and stride length were increased, while stride time and double support time were decreased. The step width was the only parameter that did not change, which could be explained by the relationship between pain and gait performance. When the pain decreased, patients with PF were able to put more weight on the symptomatic foot; therefore, the single support time was increased and the total double support time was decreased. In addition, pain reduction may reduce stride time and result in increased gait speed and cadence.
The intrinsic and extrinsic foot muscles play an important role in maintaining the arches of the foot while walking [30]. The strengthening exercise program for these muscles was expected to have better improvements in pain and gait parameters than the stretching exercise program [19,35]. However, the present study found the same effect from both programs on pain and gait. In patients with PF, there were reports about the weakness of the ankle dorsiflexors [19,36] and limitations of the ankle dorsiflexion range of motion or tightness of the ankle plantar flexors [37,38]. Any improvement may have been due to the major benefits of stretching exercise in the first month of intervention, whereas the strengthening exercises created long-term benefits at the 2-month followup. Moreover, the improvements in both groups may have occurred due to the increased ankle dorsiflexion range of motion obtained through the stretching exercises for the calf muscles and Achilles tendon [21]. In the strengthening exercises, an additional benefit was gained from the heel raise exercise, which required the utilization of forth-back muscle work to control up and down body movement while bearing body weight, improving the strength of both the ankle dorsiflexors and the plantar flexors. In addition, it also increased the ankle dorsiflexion range of motion and stretched the calf and Achilles tendons as the patients lowered down.
Further studies may examine others clinical characteristic such as the calf muscle, Achilles tendon, and plantar fascia tightness, or ankle and toe muscle strength. These clinical characteristics may affect the effectiveness of the exercise program; hence, they should be controlled to reduce the effect of these confounding factors.
The limitations of this study include the following; first, there was not a non-intervention group in this study; hence, it cannot quantify the spontaneous progression of the symptoms. Second, the study did not differentiate between bilateral or unilateral types of PF. The findings may be different due to the adaptation to these conditions, in particular, the temporospatial parameters. However, the baseline characteristics of the participants were not different because of the random allocation and control. Thirdly, confounding factors may have occurred during the study because we did not note the patients’ behaviors such as changes in activity or lifestyle, shoes, or weight. However, we asked about other interventions, exercises, and drug usage at the end of the study. Furthermore, we requested that patients avoid taking painkiller or antiinflammatory drugs, performing other foot exercises, and walking on uneven or hard surfaces. We also encouraged the patients to wear proper shoes with firm insoles and to avoid high heels. Lastly, to obtain insightful details, a long-term follow-up on these patients should occur in the future.
In conclusion, both the strengthening and stretching exercise programs could reduce the pain and improve gait performance in patients with PF within 3 months. There were no differences in the testing parameters between the strengthening and stretching groups.
ACKNOWLEDGMENTS
This work was funded by the Thailand Research Fund and Mahidol University. We would like to thank all participants, physical therapists, and staff members who assisted in this study.
AUTHOR CONTRIBUTION
Conceptualization: Thong-On S, Bovonsunthonchai S, Vachalathiti R, Smith R. Methodology: Thong-On S, Bovonsunthonchai S, Intiravoranont W, Suwannarat S. Formal analysis: Thong-On S, Bovonsunthonchai S. Funding acquisition: Bovonsunthonchai S. Visualization: Thong-On S, Bovonsunthonchai S. Writing-original draft: Thong-On S, Bovonsunthonchai S. Writing-review and editing: Thong-On S, Bovonsunthonchai S. Approval of final manuscript: all authors.
Fig. 2.
The worst pain (A) and morning pain (B) over 3 months in the strengthening (STT) and stretching (STC) groups.

Fig. 3.
The temporospatial gait parameters over 3 months in the strengthening (STT) and stretching (STC) groups: (A) cadence, (B) step width, (C) stride length, (D) stride time, (E) total double support, and (F) velocity.
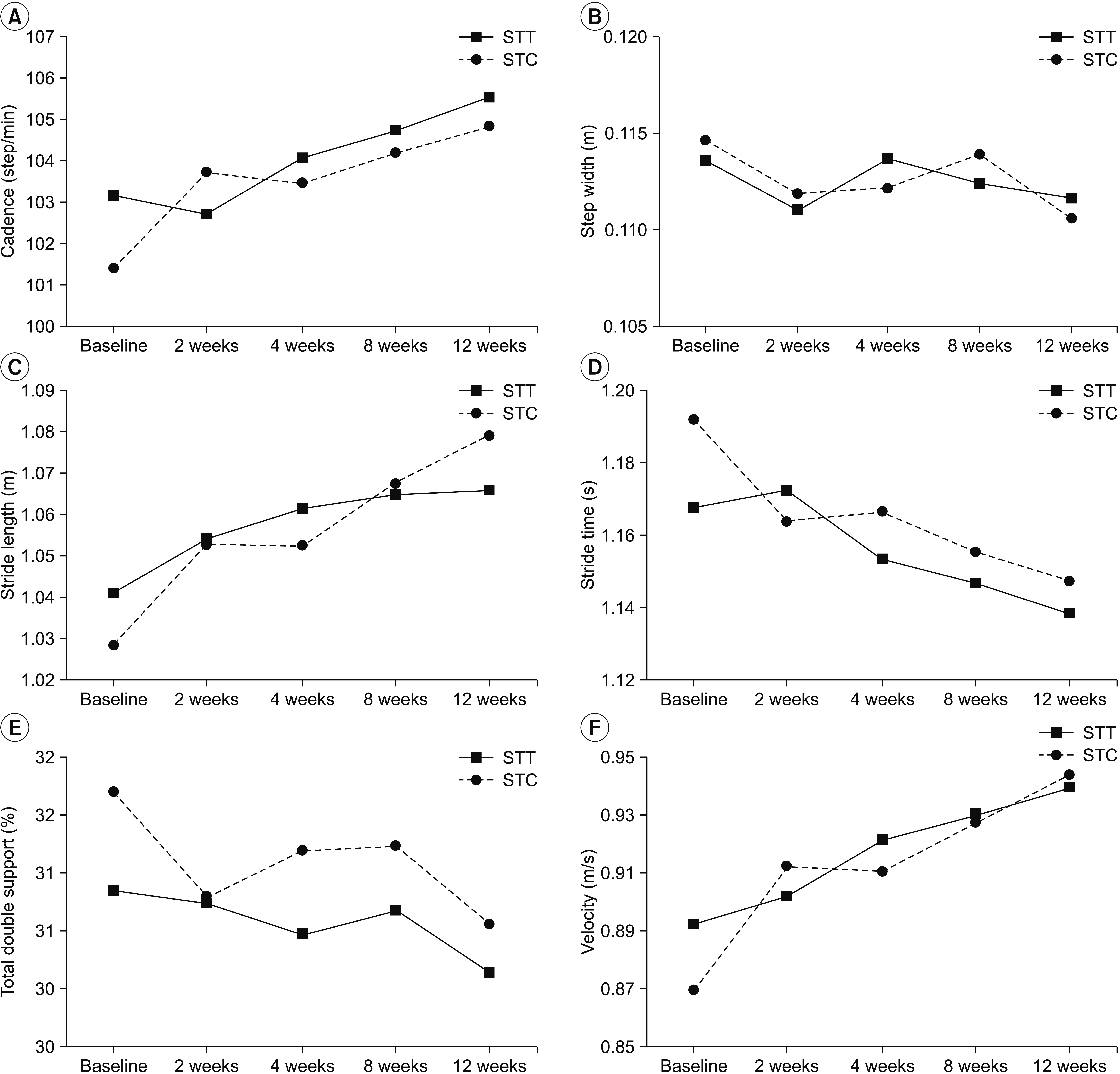
Table 1.
Manual physical therapy, strengthening exercise, and stretching exercise programs
Table 2.
Demographic data of the participants in strengthening and stretching groups
| Characteristic | Strengthening group (n=42) | Stretching group (n=42) | p-value |
|---|---|---|---|
| Age (yr)a) | 51.95±10.10 | 52.86±9.84 | 0.679 |
| Genderb) | 0.321 | ||
| Male | 13 (31) | 9 (21.4) | |
| Female | 29 (69) | 33 (78.6) | |
| Weight (kg)a) | 65.66±11.26 | 65.14±12.52 | 0.844 |
| Height (cm)a) | 161.13±9.18 | 159.45±8.13 | 0.380 |
| Pain intensity (VAS score)a) | |||
| Pain at worst | 5.96±2.30 | 5.74±2.30 | 0.660 |
| Pain at morning | 5.86±2.51 | 5.04±2.35 | 0.128 |
| PF sideb) | 1.000 | ||
| Right | 25 (59.5) | 25 (59.5) | |
| Left | 17 (40.5) | 17 (40.5) | |
| PF characteristicb) | 1.000 | ||
| Unilateral | 23 (54.8) | 23 (54.8) | |
| Bilateral | 19 (45.2) | 19 (45.2) | |
| Pain characteristicb) | |||
| Few step in the morning | 42 (100) | 40 (95.24) | 0.152 |
| After prolong sitting | 23 (54.76) | 29 (69.05) | 0.178 |
| After prolong standing or walking | 20 (47.62) | 17 (40.48) | 0.510 |
| During running | 12 (28.57) | 12 (28.57) | 1.000 |
| Underlying diseaseb) | |||
| None | 18 (42.86) | 25 (59.52) | 0.127 |
| Hypertension | 11 (26.19) | 9 (21.43) | 0.608 |
| Diabetes mellitus | 2 (4.76) | 1 (2.38) | 0.557 |
| Hyperlipidemia | 5 (11.90) | 2 (4.76) | 0.236 |
| Hyperthyroid | 3 (7.14) | 1 (2.38) | 0.306 |
| Allergy | 1 (2.38) | 3 (7.14) | 0.306 |
Table 3.
Intra- and inter-group comparisons for the pain intensity and the temporospatial gait parameters
| Parameter | Baseline | Intermediate of intervention | End of intervention | 1st month follow-up | 2nd month follow-up | df | dferror | F | p-valuea) |
|---|---|---|---|---|---|---|---|---|---|
| Worst pain (score) | |||||||||
| Strengthening group | 5.96±2.30 | 3.91±2.38 | 3.09±2.25 | 2.52±2.55 | 1.73±2.16 | 4 | 164 | 36.280 | <0.001* |
| Stretching group | 5.74±2.30 | 4.08±2.29 | 3.34±2.47 | 2.69±2.55 | 2.05±2.24 | 3.133 | 128.462 | 29.527 | <0.001* |
| t value | 0.441 | 0.341 | 0.485 | 0.317 | 0.659 | ||||
| p-valueb) | 0.660 | 0.734 | 0.629 | 0.752 | 0.512 | ||||
| Morning pain (score) | |||||||||
| Strengthening group | 5.86±2.51 | 3.70±2.28 | 2.90±2.46 | 2.25±2.51 | 1.30±1.54 | 2.812 | 115.290 | 58.565 | <0.001* |
| Stretching group | 5.04±2.35 | 3.63±2.54 | 2.75±2.52 | 1.98±2.24 | 1.52±2.12 | 2.675 | 109.672 | 32.411 | <0.001* |
| t value | 1.538 | 0.140 | 0.263 | 0.523 | 0.565 | ||||
| p-valueb) | 0.128 | 0.889 | 0.793 | 0.602 | 0.574 | ||||
| Cadence (step/min) | |||||||||
| Strengthening group | 103.16±7.75 | 102.85±8.54 | 104.03±8.42 | 104.72±8.45 | 105.50±8.47 | 2.905 | 119.114 | 3.741 | 0.014* |
| Stretching group | 101.37±10.44 | 103.70±9.82 | 103.46±10.42 | 104.16±9.35 | 104.81±9.49 | 2.764 | 113.330 | 5.301 | 0.002* |
| t value | 0.893 | 0.423 | 0.279 | 0.285 | 0.350 | ||||
| p-valueb) | 0.374 | 0.674 | 0.781 | 0.778 | 0.727 | ||||
| Step width (m) | |||||||||
| Strengthening group | 0.11±0.03 | 0.11±0.03 | 0.11±0.03 | 0.11±0.03 | 0.11±0.03 | 3.358 | 137.697 | 0.754 | 0.536 |
| Stretching group | 0.11±0.04 | 0.11±0.04 | 0.11±0.04 | 0.11±0.04 | 0.11±0.04 | 4 | 164 | 1.187 | 0.319 |
| t value | 0.118 | 0.176 | 0.278 | 0.234 | 0.123 | ||||
| p-valueb) | 0.907 | 0.861 | 0.782 | 0.816 | 0.902 | ||||
| Stride length (m) | |||||||||
| Strengthening group | 1.05±0.11 | 1.06±0.10 | 1.05±0.10 | 1.07±0.09 | 1.07±0.10 | 3.059 | 125.427 | 1.648 | 0.181 |
| Stretching group | 1.03±0.10 | 1.06±0.10 | 1.05±0.10 | 1.07±0.11 | 1.08±0.10 | 4 | 114.691 | 9.635 | <0.001* |
| t value | 0.864 | 0.293 | 0.522 | 0.098 | 0.585 | ||||
| p-valueb) | 0.390 | 0.770 | 0.603 | 0.922 | 0.560 | ||||
| Stride time (s) | |||||||||
| Strengthening group | 1.17±0.09 | 1.17±0.10 | 1.15±0.10 | 1.15±0.10 | 1.14±0.10 | 2.846 | 116.692 | 4.248 | 0.008* |
| Stretching group | 1.19±0.13 | 1.16±0.12 | 1.17±0.13 | 1.16±0.11 | 1.15±0.11 | 2.546 | 104.393 | 5.755 | 0.002* |
| t value | 0.971 | 0.299 | 0.530 | 0.411 | 0.377 | ||||
| p-valueb) | 0.334 | 0.766 | 0.598 | 0.682 | 0.707 | ||||
| Total double support (%) | |||||||||
| Strengthening group | 30.75±3.76 | 30.66±3.63 | 30.41±3.90 | 30.68±3.96 | 30.13±3.71 | 3.264 | 133.840 | 1.343 | 0.262 |
| Stretching group | 31.69±3.52 | 30.79±3.61 | 31.18±3.72 | 31.23±3.95 | 30.55±3.24 | 4 | 164 | 2.856 | 0.025* |
| t value | 1.187 | 0.167 | 0.955 | 0.635 | 0.544 | ||||
| p-valueb) | 0.239 | 0.868 | 0.342 | 0.527 | 0.588 | ||||
| Gait speed (m/s) | |||||||||
| Strengthening group | 0.90±0.11 | 0.91±0.12 | 0.92±0.12 | 0.93±0.12 | 0.94±0.12 | 3.264 | 133.840 | 1.343 | 0.262 |
| Stretching group | 0.87±0.13 | 0.91±0.13 | 0.91±0.13 | 0.93±0.13 | 0.94±0.13 | 2.680 | 109.868 | 11.772 | <0.001* |
| t value | 1.071 | 0.176 | 0.507 | 0.171 | 0.141 | ||||
| p-valueb) | 0.287 | 0.861 | 0.641 | 0.864 | 0.888 |
Table 4.
Pairwise comparison for the strengthening group (p-value)
| Parameter | Baseline | Intermediate of intervention | End of intervention | 1st month follow-up | 2nd month follow-up |
|---|---|---|---|---|---|
| Worst pain (VAS score) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | <0.001* | N/A | |||
| End of intervention | <0.001* | 0.107 | N/A | ||
| 1st month follow-up | <0.001* | 0.002* | 0.877 | N/A | |
| 2nd month follow-up | <0.001* | <0.001* | 0.005* | 0.261 | N/A |
| Morning pain (VAS score) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | <0.001* | N/A | |||
| End of intervention | <0.001* | 0.028* | N/A | ||
| 1st month follow-up | <0.001* | <0.001* | 0.074 | N/A | |
| 2nd month follow-up | <0.001* | <0.001* | <0.001* | 0.002* | N/A |
| Cadence (step/min) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | 1.000 | N/A | |||
| End of intervention | 1.000 | 0.731 | N/A | ||
| 1st month follow-up | 1.000 | 0.052 | 1.000 | N/A | |
| 2nd month follow-up | 0.296 | 0.031* | 0.759 | 1.000 | N/A |
| Stride length (m) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | 0.776 | N/A | |||
| End of intervention | 0.747 | 1.000 | N/A | ||
| 1st month follow-up | 0.768 | 1.000 | 1.000 | N/A | |
| 2nd month follow-up | 0.856 | 1.000 | 1.000 | 1.000 | N/A |
| Stride time (s) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | 1.000 | N/A | |||
| End of intervention | 1.000 | 0.215 | N/A | ||
| 1st month follow-up | 0.749 | 0.018* | 1.000 | N/A | |
| 2nd month follow-up | 0.255 | 0.020* | 1.000 | 1.000 | N/A |
| Total double support (%) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | 1.000 | N/A | |||
| End of intervention | 1.000 | 1.000 | N/A | ||
| 1st month follow-up | 1.000 | 1.000 | 1.000 | N/A | |
| 2nd month follow-up | 0.928 | 1.000 | 1.000 | 0.785 | N/A |
| Gait speed (m/s) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | 1.000 | N/A | |||
| End of intervention | 1.000 | 1.000 | N/A | ||
| 1st month follow-up | 1.000 | 1.000 | 1.000 | N/A | |
| 2nd month follow-up | 0.928 | 1.000 | 1.000 | 0.785 | N/A |
Table 5.
Pairwise comparison for the stretching group (p-value)
| Parameter | Baseline | Intermediate of intervention | End of intervention | 1st month follow-up | 2nd month follow-up |
|---|---|---|---|---|---|
| Worst pain (VAS score) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | 0.002* | N/A | |||
| End of intervention | <0.001* | 0.095 | N/A | ||
| 1st month follow-up | <0.001* | 0.004* | 0.434 | N/A | |
| 2nd month follow-up | <0.001* | <0.001* | 0.005* | 0.246 | N/A |
| Morning pain (VAS score) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | 0.011* | N/A | |||
| End of intervention | <0.001* | 0.001* | N/A | ||
| 1st month follow-up | <0.001* | <0.001* | 0.043* | N/A | |
| 2nd month follow-up | <0.001* | <0.001* | 0.016* | 0.569 | N/A |
| Cadence (step/min) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | 0.049 | N/A | |||
| End of intervention | 0.287 | 1.000 | N/A | ||
| 1st month follow-up | 0.012* | 1.000 | 1.000 | N/A | |
| 2nd month follow-up | 0.045* | 1.000 | 0.935 | 1.000 | N/A |
| Stride length (m) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | 0.047* | N/A | |||
| End of intervention | 0.126 | 1.000 | N/A | ||
| 1st month follow-up | 0.002* | 1.000 | 0.433 | N/A | |
| 2nd month follow-up | 0.001* | 0.136 | 0.032 | 0.746 | N/A |
| Stride time (s) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | 0.106 | N/A | |||
| End of intervention | 0.384 | 1.000 | N/A | ||
| 1st month follow-up | 0.008* | 1.000 | 1.000 | N/A | |
| 2nd month follow-up | 0.043* | 0.757 | 0.540 | 1.000 | N/A |
| Total double support (%) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | 0.258 | N/A | |||
| End of intervention | 1.000 | 1.000 | N/A | ||
| 1st month follow-up | 1.000 | 1..000 | 1.000 | N/A | |
| 2nd month follow-up | 0.030* | 1.000 | 0.629 | 0.730 | N/A |
| Gait speed (m/s) | |||||
| Baseline | N/A | ||||
| Intermediate of intervention | 0.010* | N/A | |||
| End of intervention | 0.056 | 1.000 | N/A | ||
| 1st month follow-up | 0.001* | 1.000 | 0.279 | N/A | |
| 2nd month follow-up | <0.001* | 0.075 | 0.003* | 0.7002 | N/A |
REFERENCES
1. McPoil TG, Martin RL, Cornwall MW, Wukich DK, Irrgang JJ, Godges JJ. Heel pain: plantar fasciitis: clinical practice guidelines linked to the international classification of function, disability, and health from the orthopaedic section of the American Physical Therapy Association. J Orthop Sports Phys Ther 2008;38:A1-18.

2. Cole C, Seto C, Gazewood J. Plantar fasciitis: evidence-based review of diagnosis and therapy. Am Fam Physician 2005;72:2237-42.

3. Goff JD, Crawford R. Diagnosis and treatment of plantar fasciitis. Am Fam Physician 2011;84:676-82.

4. Martin RL, Davenport TE, Reischl SF, McPoil TG, Matheson JW, Wukich DK, et al. Heel pain: plantar fasciitis: revision 2014. J Orthop Sports Phys Ther 2014;44:A1-33.


5. Pfeffer G, Bacchetti P, Deland J, Lewis A, Anderson R, Davis W, et al. Comparison of custom and prefabricated orthoses in the initial treatment of proximal plantar fasciitis. Foot Ankle Int 1999;20:214-21.


6. Johnson RE, Haas K, Lindow K, Shields R. Plantar fasciitis: what is the diagnosis and treatment? Orthop Nurs 2014;33:198-204.


7. McMillan AM, Landorf KB, Barrett JT, Menz HB, Bird AR. Diagnostic imaging for chronic plantar heel pain: a systematic review and meta-analysis. J Foot Ankle Res 2009;2:32.



8. Healey K, Chen K. Plantar fasciitis: current diagnostic modalities and treatments. Clin Podiatr Med Surg 2010;27:369-80.


9. Irving DB, Cook JL, Young MA, Menz HB. Impact of chronic plantar heel pain on health-related quality of life. J Am Podiatr Med Assoc 2008;98:283-9.


10. Chang R, Rodrigues PA, Van Emmerik RE, Hamill J. Multi-segment foot kinematics and ground reaction forces during gait of individuals with plantar fasciitis. J Biomech 2014;47:2571-7.


11. Yoo SD, Kim HS, Lee JH, Yun DH, Kim DH, Chon J, et al. Biomechanical parameters in plantar fasciitis measured by gait analysis system with pressure sensor. Ann Rehabil Med 2017;41:979-89.




12. Hsu WH, Lai LJ, Chang HY, Hsu RW. Effect of shockwave therapy on plantar fasciopathy: a biomechanical prospective. Bone Joint J 2013;95-B:1088-93.


15. Digiovanni BF, Nawoczenski DA, Malay DP, Graci PA, Williams TT, Wilding GE, et al. Plantar fascia-specific stretching exercise improves outcomes in patients with chronic plantar fasciitis: a prospective clinical trial with two-year follow-up. J Bone Joint Surg Am 2006;88:1775-81.


16. Thompson JV, Saini SS, Reb CW, Daniel JN. Diagnosis and management of plantar fasciitis. J Am Osteopath Assoc 2014;114:900-6.


17. Pohl MB, Hamill J, Davis IS. Biomechanical and anatomic factors associated with a history of plantar fasciitis in female runners. Clin J Sport Med 2009;19:372-6.


18. Ajimsha MS, Binsu D, Chithra S. Effectiveness of myofascial release in the management of plantar heel pain: a randomized controlled trial. Foot (Edinb) 2014;24:66-71.


19. Sullivan J, Burns J, Adams R, Pappas E, Crosbie J. Musculoskeletal and activity-related factors associated with plantar heel pain. Foot Ankle Int 2015;36:37-45.


20. Chang R, Kent-Braun JA, Hamill J. Use of MRI for volume estimation of tibialis posterior and plantar intrinsic foot muscles in healthy and chronic plantar fasciitis limbs. Clin Biomech (Bristol, Avon) 2012;27:500-5.


21. Rathleff MS, Molgaard CM, Fredberg U, Kaalund S, Andersen KB, Jensen TT, et al. High-load strength training improves outcome in patients with plantar fasciitis: a randomized controlled trial with 12-month follow-up. Scand J Med Sci Sports 2015;25:e292. -300.


22. Renan-Ordine R, Alburquerque-Sendin F, de Souza DP, Cleland JA, Fernandez-de-Las-Penas C. Effectiveness of myofascial trigger point manual therapy combined with a self-stretching protocol for the management of plantar heel pain: a randomized controlled trial. J Orthop Sports Phys Ther 2011;41:43-50.


23. Karabay N, Toros T, Hurel C. Ultrasonographic evaluation in plantar fasciitis. J Foot Ankle Surg 2007;46:442-6.


24. Grecco MV, Brech GC, Greve JM. One-year treatment follow-up of plantar fasciitis: radial shockwaves vs. conventional physiotherapy. Clinics (Sao Paulo) 2013;68:1089-95.




25. Cleland JA, Abbott JH, Kidd MO, Stockwell S, Cheney S, Gerrard DF, et al. Manual physical therapy and exercise versus electrophysical agents and exercise in the management of plantar heel pain: a multicenter randomized clinical trial. J Orthop Sports Phys Ther 2009;39:573-85.


26. Shashua A, Flechter S, Avidan L, Ofir D, Melayev A, Kalichman L. The effect of additional ankle and midfoot mobilizations on plantar fasciitis: a randomized controlled trial. J Orthop Sports Phys Ther 2015;45:265-72.


27. DiGiovanni BF, Nawoczenski DA, Lintal ME, Moore EA, Murray JC, Wilding GE, et al. Tissue-specific plantar fascia-stretching exercise enhances outcomes in patients with chronic heel pain: a prospective, randomized study. J Bone Joint Surg Am 2003;85:1270-7.


28. Cinar E, Saxena S, Uygur F. Low-level laser therapy in the management of plantar fasciitis: a randomized controlled trial. Lasers Med Sci 2018;33:949-58.



29. Sweeting D, Parish B, Hooper L, Chester R. The effectiveness of manual stretching in the treatment of plantar heel pain: a systematic review. J Foot Ankle Res 2011;4:19.




30. Kamonseki DH, Goncalves GA, Yi LC, Junior IL. Effect of stretching with and without muscle strengthening exercises for the foot and hip in patients with plantar fasciitis: a randomized controlled single-blind clinical trial. Man Ther 2016;23:76-82.


31. Lee JS, Hobden E, Stiell IG, Wells GA. Clinically important change in the visual analog scale after adequate pain control. Acad Emerg Med 2003;10:1128-30.


32. Landorf KB, Radford JA, Hudson S. Minimal Important Difference (MID) of two commonly used outcome measures for foot problems. J Foot Ankle Res 2010;3:7.




33. Brach JS, Perera S, Studenski S, Katz M, Hall C, Verghese J. Meaningful change in measures of gait variability in older adults. Gait Posture 2010;31:175-9.


34. Oberg T, Karsznia A, Oberg K. Basic gait parameters: reference data for normal subjects, 10-79 years of age. J Rehabil Res Dev 1993;30:210-23.

35. Mulligan EP, Cook PG. Effect of plantar intrinsic muscle training on medial longitudinal arch morphology and dynamic function. Man Ther 2013;18:425-30.


36. Kibler WB, Goldberg C, Chandler TJ. Functional biomechanical deficits in running athletes with plantar fasciitis. Am J Sports Med 1991;19:66-71.










