- Search
| Ann Rehabil Med > Volume 37(1); 2013 > Article |
Abstract
Objective
To determine the useful tool for evaluating salivary aspiration in brain-injured patients with tracheostomy.
Methods
Radionuclide salivagram and laryngoscopy was done in 27 brain-injured patients with tracheostomy. During salivagram, 99mTc sulfur colloid was placed sublingually in the supine position, and 50-minute dynamic images and 2-hour delayed images were obtained. Salivary aspiration was detected when the tracer was entered into the major airways or lung parenchyma. Laryngoscopy was done by otolaryngologists, and saliva aspiration, saliva pooling, and vocal cord palsy were evaluated. Videofluoroscopic swallowing study was done in patients who were able to undergo the test.
Results
The detection rate of salivary aspiration was 44.4% with salivagram, and 29.6% with laryngoscopy. The correlation of the two tests was 70.4%. Of the laryngoscopy findings, salivary pooling had significant correlation with positive salivagram results (p=0.04). Frequent need of suction correlated with salivary aspiration in both salivagram (p=0.01) and laryngoscopy (p=0.01). Patients with negative results in salivagram or laryngoscopy had higher rates of progressing to oral feeding or tapering tracheostomy. Two patients developed aspiration pneumonia, and both patients only showed aspiration in salivagram.
Conclusion
Brain-injured patients with tracheostomy have a high risk of salivary aspiration. Evaluation of salivary aspiration is important, as it may predict aspiration pneumonia and aids in clinical decisions of oral feeding or tracheostomy removal. Salivagram is more sensitive than laryngoscopy, but laryngoscopy may be useful for evaluating structural abnormalities or for follow-up examinations to assess the changes.
Pulmonary aspiration is a common problem in patients with brain injury, and it can cause purulent sputum and pneumonia. Causes of pulmonary aspiration include aspiration due to swallowing dysfunction, aspiration from gastroesophageal reflux, and salivary aspiration. The last of these is the least commonly recognized form of aspiration, but can result in recurrent pneumonia from pathogenic bacteria and yeast in the oral cavity [1]. Risk factors of salivary aspiration include swallowing incoordination, absent laryngeal sensation, excess production of saliva, and vocal cord paresis. Videofluroscopic swallowing studies (VFSS), scintigraphic swallowing studies, and fiberoptic endoscopic evaluation of swallowing are used to assess food aspiration. For salivary aspiration, radionuclide salivagram and laryngoscopy (fiberoptic endoscopy) can be used, but a standard test has not been established [2].
Heyman and Respondek [3] and Heyman [4] first described radionuclide salivagrams in 1989; a small dose of radiotracer is injected into the patient's mouth, and serial images are recorded. Salivary aspiration is diagnosed when tracheal or bronchial activity is detected. Previous studies claim that salivagrams are the most sensitive test for saliva aspiration [5]. However, it is used mostly in children, and there are few studies that have assessed its utility in adults. Laryngoscopy can be used to evaluate salivary aspiration or pooling, vocal cord abnormality, and other relevant structures. Pooled oral secretions may predict aspiration, but the study is done in a short period of time and may lack sensitivity [6].
Patients with tracheostomy are at a high risk for aspiration, which can occur for various reasons, such as pharyngeal pooling of secretions above the airway cuff, decreased laryngeal elevation, desensitization of the larynx, and loss of protective reflexes. The incidence of aspiration in adults with tracheostomy is reported to be 30% to 50%, and the risk of aspiration is directly related to the amount of oropharyngeal secretion [7,8]. Also, pneumonia rates were higher even in tube feeding patients, whom food aspiration is less likely to occur, indicating a possibility of pneumonia from salivary aspiration [8]. These findings underscore the need to evaluate salivary aspiration in patients with tracheostomy, followed with appropriate management to prevent pneumonia in high risk patients.
The goal of this study is to compare the two tools-salivagram and laryngoscopy, for evaluating salivary aspiration, and also to explore the clinical significance by investigating the frequency and associated factors of salivary aspiration in brain-injured patients with tracheostomy.
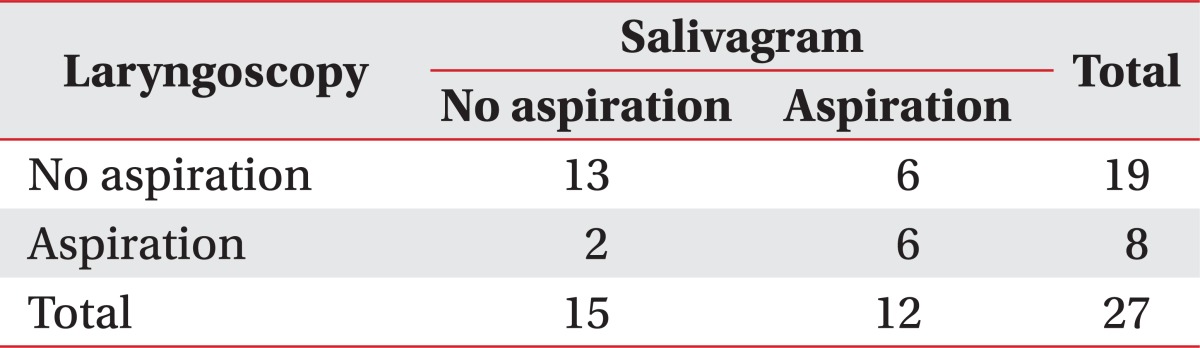
A total of 27 brain-injured patients with tracheostomy tubes, who were transferred or admitted to the rehabilitation department between April 2011 and December 2011, were included in the study. We enrolled only patients who required suction of sputum or saliva. Radionuclide salivagram and laryngoscopy were conducted with an interval of less than 3 days. Patients who were medically unstable or unable to follow simple one step commands were excluded.
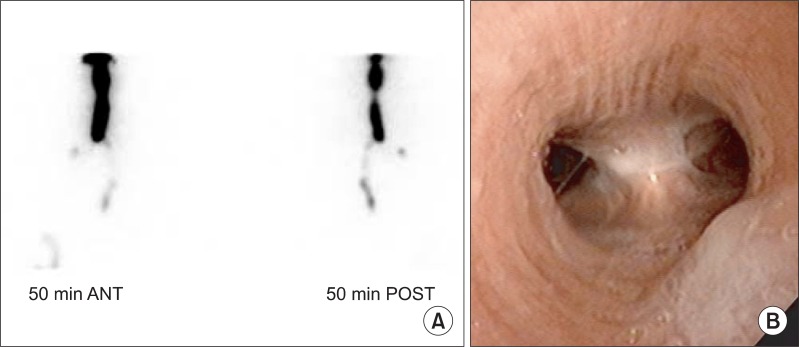
For radionuclide salivagram, all patients fasted for 2 hours before the test. The patient was placed in a supine position under a gamma camera fitted with a low-energy high-resolution collimator and received a sublingual drooping of 0.5 mL saline with 0.5 mCi Tc-99m diethylene triamine pentaacetic acid (18.5 MBq). Dynamic images of the mouth to the upper abdomen were recorded for 50 minutes, and after, anteroposterior and lateral static chest images were also obtained. Any suggestive findings of positive radionuclide salivagram from these images were an indication of ending the test. In case of negative or inconclusive findings on the initial 50-minute test, delayed anteroposterior and lateral static chest images were acquired after 2 hours. Interpretation of the images was performed by two expertise nuclear medicine physicians. Salivary aspiration was indicated by detection of positive radioactivity in dynamic and/or static images on tracheobronchial tree, either side of both lung fields or site of tracheostomy (Fig. 1A).
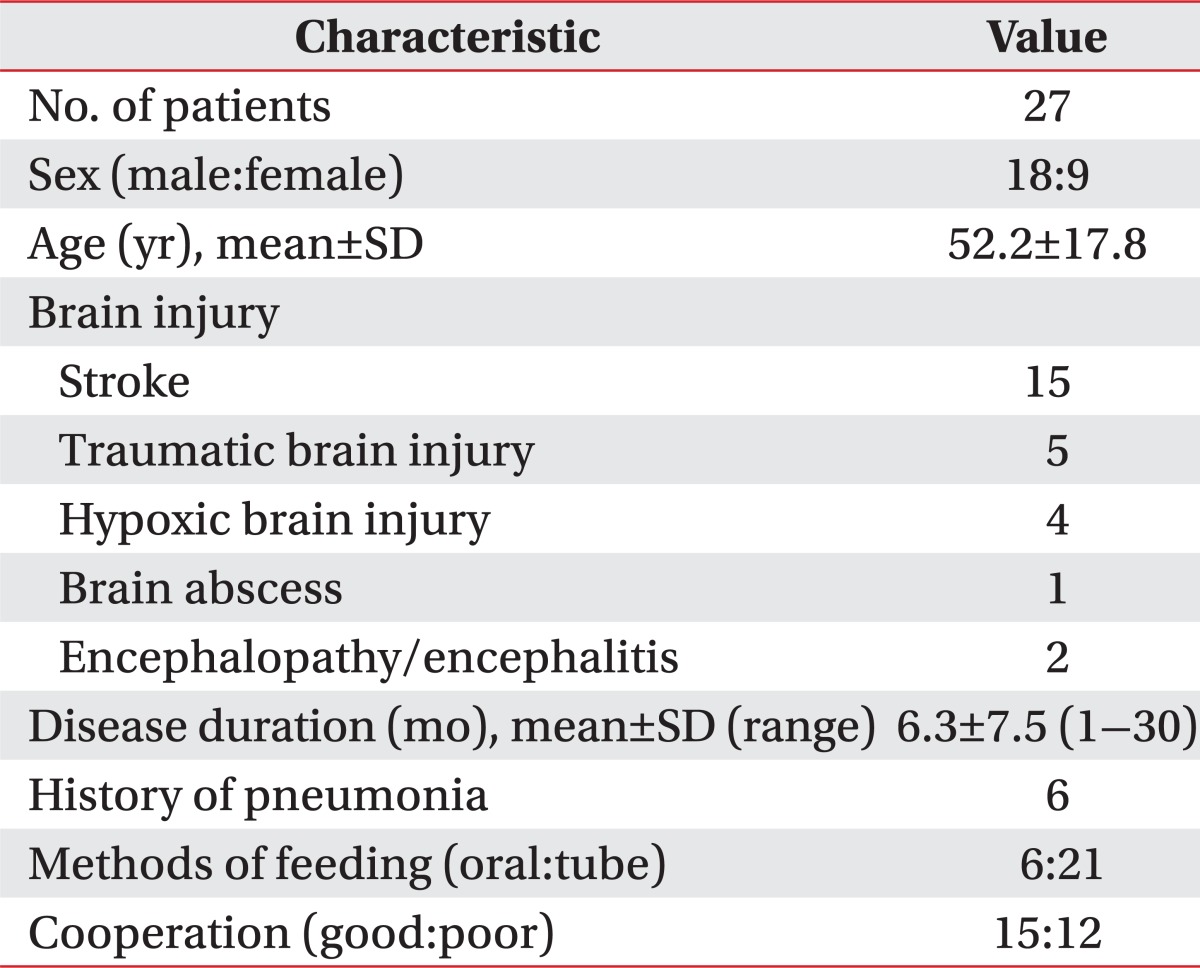
Fiberoptic laryngoscopy was performed by an otolaryngologist through the tracheostomy. During the procedure, salivary aspiration, pooling of secretions in the valleculae or pyrifrom sinuses, vocal cord gap and mobility, and other airway structures were evaluated. Salivary aspiration was assessed when saliva in the trachea or active aspiration was visualized (Fig. 1B). Structures evaluated included the vallecula, epiglottis, arytenoids, true and false vocal cords, and the oral and nasal cavity.
Disease duration, history of pneumonia, cooperation level, and feeding method were assessed. Cooperation level was evaluated by the ability to participate in a Mini-Mental Status Examination-Korean version. VFSS was done in patients who were able to perform the protocol. The clinical course of the patients was followed until the patient was discharged from the hospital.
Data analysis was performed using SPSS ver. 18.0. (SPSS Inc., Chicago, IL, USA) correlation between variables were determined by Žć2 analysis with the Fisher's exact test. Significance was defined as p<0.05. The study was approved by the Institutional Review Board.
Twenty-seven patients who were enrolled completed the evaluations, and their demographic data are presented in Table 1. All patients who were able to follow simple commands were evaluated with both laryngoscopy and salivagram. The studies were conducted within intervals of 3 days, and all patients were medically stable at the time of the study.
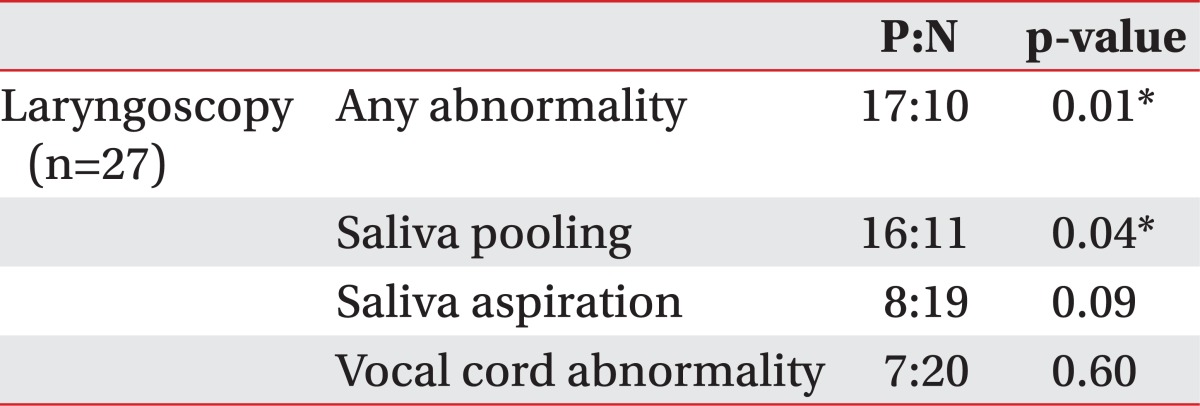
Salivary aspiration was detected in 12 out of 27 patients with salivagram. On laryngoscopy, 8 out of 27 patients showed salivary aspiration. Among the 8 patients who showed aspiration, 6 patients also had aspiration in the salivagram (Table 2). The frequency of salivary aspiration among patients was 44.4% with salivagram and 29.6% with laryngoscopy, and the correlation between the two studies was 70.4% (p=0.09 by Fisher's exact test).
The association between laryngoscopy findings and salivagram results are shown in Table 3. There was a significant association between any abnormality, including saliva pooling, saliva aspiration, or vocal cord abnormality, and salivary pooling observed by laryngoscopy with salivary aspiration on salivagram. Salivary aspiration or vocal cord abnormality seen on a laryngoscopy did not show significant association with salivary aspiration on salivagram.
The association of clinical characteristics with salivagram and laryngoscopy is presented in Table 4. Of the clinical characteristics, frequent manual suction of secretions (more than ten times per day) was significantly associated with salivary aspiration seen on both salivagram and laryngoscopy.
Among the 27 patients, only 15 patients could be evaluated by VFSS; the others were unable or unwilling to cooperate. Of the 15 patients who completed the study, five showed aspiration, five showed penetration, and five had no abnormalities. We compared the salivagram results with those of VFSS and found that of the 12 patients who were diagnosed with salivary aspiration by salivagram, 4 patients also had aspiration in VFSS, 2 patients had penetration, and the other 6 patients failed the study or could not be evaluated. In comparing the laryngoscopy and VFSS results, of the 8 patients who had aspiration as assessed by laryngoscopy, 4 also had aspiration in VFSS, 2 had penetration, and the others could not be tested.
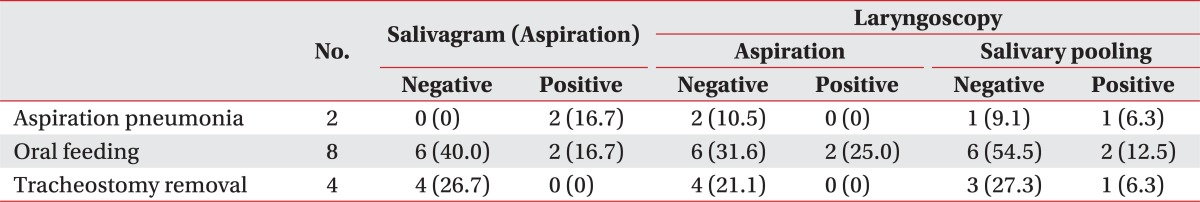
Table 5 shows the clinical course of patients according to the findings on salivagram or laryngoscopy. The follow-up period of patients ranged from 1 to 3 months (mean, 1.7 months). Of the 12 patients who showed aspiration on salivagram, only 2 were able to progress to oral feeding, compared to 6 in patients without aspiration. None of the patients with aspiration on salivagram were able to remove tracheostomy, while four patients without aspiration had their tracheostomy closed. Similar results were seen in patients with positive laryngoscopy results. Two patients developed aspiration pneumonia, and these patients both showed aspiration on salivagram, whereas in laryngoscopy, one showed only salivary pooling, and one had no abnormality.
In this study, we found that the frequency of salivary aspiration in brain-injured patients with tracheostomy was as high as 44.4% by salivagram. However, the frequency was lower when evaluated with laryngoscopy, which demonstrated salivary aspiration in 29.6% of patients. Both tests were used to evaluate salivary aspiration in patients who were unable to cooperate during VFSS.
This is the first study to evaluate salivary aspiration using both laryngoscopy and salivagram.
To date, salivagram is most commonly used in children who cannot cooperate during VFSS to evaluate aspiration. Since Heyman and Respondek [3] and Heyman [4] first proposed the use of salivagram for detecting aspiration in children, previous studies have mostly involved children. In a study that compared salivagram in severe cerebral palsy (CP), who are at high risk of aspiration with able-bodied adult volunteers, the frequency of aspiration was 56% in children with CP compared to 0% in young adults, showing that salivagram is a useful tool for the demonstration of aspiration [9]. Other studies have reported the frequency of positive salivagram in high risk children as 26% to 28% [10,11]. The difference in the rate of aspiration can be explained by the different infusion method; the former study used a continuous infusion method for 1 hour, whereas the latter studies used the sublingual dose method, as in our study. When Kang et al. [2] studied bedridden patients with brain lesions, the frequency of positive response was 22%. Silver and Van Nostrand [12] reported a case series of patients evaluated with salivagram, in which one patient had tracheostomy. These are the only studies to our knowledge that evaluated adults at risk of aspiration with salivagram. The frequency of salivary aspiration, detected by salivagram in our study, was higher than the previous studies. This may imply that tracheostomy further increases the risk of salivary aspiration in addition to deficits of pharyngeal or laryngeal control caused by brain lesions.
It is not clear yet whether positive salivagram results predict more frequent episodes of acute aspiration or lung disease. However, Bar-Sever et al. [10] demonstrated the utility of salivagram in assessing children with unexplained lung disease or recurrent pneumonias. Twenty-six percent showed salivary aspiration, suggesting aspiration of oral contents as a contributing factor [10]. Cook et al. [13] suggested salivagram as a tool for identifying candidates for laryngotracheal separation as a definitive mean of eliminating aspiration.
Few studies have compared salivagram with other tools for evaluating aspiration. In a study by Baikie et al. [5], positive results for aspiration in CP patients were most frequent in salivagram, compared to that of VFSS and milk scans. One explanation for this result is that aspiration is an intermittent event, and because salivagram has the longest monitoring time, it is more likely to detect aspiration.
Laryngoscopy is useful for evaluating the pharyngeal structures and can directly observe aspiration. This method can also be used to assess laryngeal sensation or to evaluate the swallowing process, as in fiberoptic endoscopic evaluation of swallowing. Other advantages of laryngoscopic evaluation includes that it can be performed bedside, it does not require fluoroscopic equipments, the patient is not exposed to radiation, and minimal positioning or cooperation of the patient is needed. It can also be repeated over time to assess the changes in a patient's swallowing function [14]. Oropharyngeal secretions, frequency of spontaneous swallows, and laryngopharyngeal sensation were highly predictive of aspiration in a study by Murray et al. [6]. Here, we focused on the detection of salivary pooling, aspiration, and structural problems. Sixty-three percent of the patients had abnormality detected by laryngoscopy. Salivary pooling was the most common problem, occurring in 59.3%.
The lower rate of salivary aspiration in laryngoscopy compared to salivagram can be explained by the shorter evaluation time. Another possible reason is patient positioning during the tests. During the salivagram, patients are supine, whereas laryngoscopy is done in an upright position, and this may increase the aspiration rate in salivagram. The results demonstrate that salivary pooling should also be considered as an important predictor for aspiration, as it showed significant association with a positive salivagram. When this is considered, laryngoscopy may be useful for assessing changes in follow-up studies, as it is more simple and convenient than salivagram.
Patients with positive salivagram results showed either aspiration or penetration on VFSS, but only half of the patients could undergo the latter study. Patients only need to follow simple commands in order to undergo salivagram, whereas more level of cooperation is needed to participate in VFSS. In those that only show penetration on VFSS who are being considered for oral feeding or removal of tracheostomy, salivagram may provide additional information to evaluate aspiration risk.
Clinical factors related to positive salivagram results in previous studies include recurrent pneumonia and gastroesophageal reflux in children and anterior drooling, and cooperation in brain-injured adults [2,15]. Our results demonstrated a significant association between frequent suction of secretions and salivary aspiration. This can easily be explained since patients who lack control and/or have copious oral secretions will need frequent manual suction, either orally or through their tracheostomy, and this also increases the risk of aspirating secretions.
The possible clinical applications of the two tests evaluating salivary aspiration can be inferred from the clinical courses of patients. Both salivagram and laryngoscopy correlated well with the progression to oral feeding and removal of tracheostomy. Negative results on salivagram and laryngoscopy may be useful for informing decisions on feeding or tracheostomy tapering. Salivagram appeared to be more helpful in predicting pneumonia, as two patients who developed aspiration pneumonia only had positive results on salivagram.
The strength of this study includes that it is the first study to evaluate salivary aspiration with both salivagram and laryngoscopy, and compare the findings. Also, we selected a homogeneous high risk group, brain-injured patients with tracheostomy, and demonstrated the frequency and clinical implications of salivary aspiration.
There are also a few limitations to our study. The duration of observation of the clinical course was limited to the inpatient rehabilitation period, and the sample size was small. Also, only a part of the subjects were evaluated by VFSS, therefore, comparison with VFSS results is limited. Further studies with larger sample size and longer follow-up period will be required to ascertain the results of our study.
In conclusion, the rate of salivary aspiration in brain-injured patients with tracheostomy as evaluated by salivagram was 44.4%. Salivagram was more sensitive than laryngoscopy in detecting aspiration. Evaluating salivary aspiration is important in brain-injured patients with tracheostomy, especially in patients with salivary pooling or frequent need of suction, and appropriate interventions should be considered according to the results.
References
1. Boesch RP, Daines C, Willging JP, Kaul A, Cohen AP, Wood RE, et al. Advances in the diagnosis and management of chronic pulmonary aspiration in children. Eur Respir J 2006;28:847-861. PMID: 17012631.


2. Kang IS, Kwon JG, Lee SU, Lee ZI, Park GY, Park HW. Detection of saliva aspiration using salivagram in bedridden patients with brain lesion. J Korean Acad Rehabil Med 2010;34:503-507.
3. Heyman S, Respondek M. Detection of pulmonary aspiration in children by radionuclide "salivagram". J Nucl Med 1989;30:697-699. PMID: 2715832.

4. Heyman S. The radionuclide salivagram for detecting the pulmonary aspiration of saliva in an infant. Pediatr Radiol 1989;19:208-209. PMID: 2497434.


5. Baikie G, South MJ, Reddihough DS, Cook DJ, Cameron DJ, Olinsky A, et al. Agreement of aspiration tests using barium videofluoroscopy, salivagram, and milk scan in children with cerebral palsy. Dev Med Child Neurol 2005;47:86-93. PMID: 15707231.


6. Murray J, Langmore SE, Ginsberg S, Dostie A. The significance of accumulated oropharyngeal secretions and swallowing frequency in predicting aspiration. Dysphagia 1996;11:99-103. PMID: 8721067.


7. Elpern EH, Jacobs ER, Bone RC. Incidence of aspiration in tracheally intubated adults. Heart Lung 1987;16:527-531. PMID: 3115917.

8. Donzelli J, Brady S, Wesling M, Theisen M. Secretions, occlusion status, and swallowing in patients with a tracheotomy tube: a descriptive study. Ear Nose Throat J 2006;85:831-834. PMID: 17240710.


9. Baikie G, Reddihough DS, South M, Cook DJ. The salivagram in severe cerebral palsy and able-bodied adults. J Paediatr Child Health 2009;45:342-345. PMID: 19490413.


10. Bar-Sever Z, Connolly LP, Treves ST. The radionuclide salivagram in children with pulmonary disease and a high risk of aspiration. Pediatr Radiol 1995;25(Suppl 1): S180-S183. PMID: 8577521.


11. Levin K, Colon A, DiPalma J, Fitzpatrick S. Using the radionuclide salivagram to detect pulmonary aspiration and esophageal dysmotility. Clin Nucl Med 1993;18:110-114. PMID: 8432052.


12. Silver KH, Van Nostrand D. Scintigraphic detection of salivary aspiration: description of a new diagnostic technique and case reports. Dysphagia 1992;7:45-49. PMID: 1424829.


13. Cook SP, Lawless S, Mandell GA, Reilly JS. The use of the salivagram in the evaluation of severe and chronic aspiration. Int J Pediatr Otorhinolaryngol 1997;41:353-361. PMID: 9350494.


14. Kaye GM, Zorowitz RD, Baredes S. Role of flexible laryngoscopy in evaluating aspiration. Ann Otol Rhinol Laryngol 1997;106:705-709. PMID: 9270438.


15. Simons JP, Rubinstein EN, Mandell DL. Clinical predictors of aspiration on radionuclide salivagrams in children. Arch Otolaryngol Head Neck Surg 2008;134:941-944. PMID: 18794438.


Fig.┬Ā1
(A) Example of a positive salivagram image shows radiotracer present in the right trachea. (B) Laryngoscopic image shows direct view of aspirated saliva in the trachea. ANT, anterior; POST, posterior.