INTRODUCTION
Pain and itching are two major long-term symptoms that occur after burn injury, reported in as high as 70% of burn survivors with large burn injuries [
1,
2]. These symptoms are significant disabilities in this population, interfering with sleep, disrupting concentration, and impacting other important quality of life elements, such as return to work [
3]. Study results have found that as high as 28% of burn patients are not employed at longterm follow-up [
4]. Furthermore, patients with severe burn injury experience multiple long-term sequelae. In a vast majority of cases, additional disorders, such as depression, post-traumatic stress syndrome, and anxiety, also emerge. Pruritus also remains a major problem in the burn population, affecting not only adults, but also children [
5,
6]. The fact that these types of injuries result in long-term impairments demonstrates the need for additional effective treatments. Through further research however, it is possible to address these unmet clinical needs. Current research on pain has begun to address the proper treatment for patients with burn injury [
1], but there remains more to be investigated.
Very little is currently known about the mechanism and biological effects of pruritus, thus there has been no effective treatment reported to date. For this reason, the current study aims to investigate cortical reorganizational changes due to the pruritus experienced by some patients with burn injury. Plastic changes in the mammalian brain have been observed following large scale injuries [
7]. Specifically, studies in patients with chronic pain have shown evident changes in cortical activity compared to the cortical activity in healthy subjects [
8,
9]. Therefore, it was hypothesized that pruritus in burn patients, similarly to chronic pain, should also lead to maladaptive brain reorganization, that causes changes in cortical activity compared to healthy subjects.
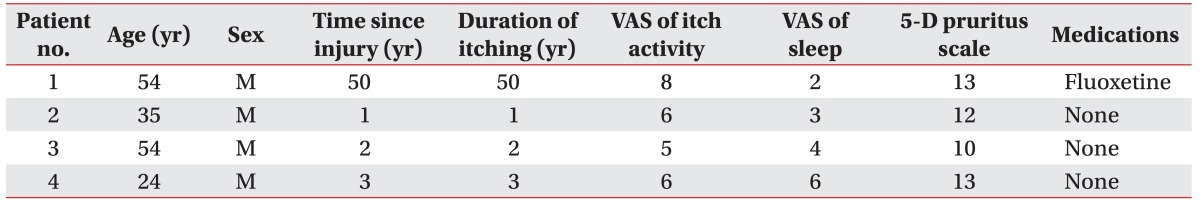
Patients more than 3 weeks from the time of their burn injury, with pruritus, including subacute and chronic pruritus, due to burn injuries were recruited for this study; each was gender and age matched to a healthy subject. Specifically, chronic pruritus was defined as itching persisting for more than 6 weeks [
10].
DISCUSSION
Chronic pruritus and pain are related to damage or pathological changes in the peripheral nervous system, though its relation to the long-term changes in central nervous system transmission is not well understood [
12]. Given the current lack of existing literature addressing this condition, this exploratory study demonstrates the need to conduct research concerning pruritus. Previous studies have provided evidence of maladaptive brain reorganization in pain by using transcranial magnetic stimulation to assess motor cortex excitability [
13]. However, in this current study, focus was placed instead on the changes in the brain waves and assessed by using the EEG.
This study employed the use of quantitative electroencephalography (qEEG) to analyze the changes in brainwave power. In particular, focus was placed on the alpha, beta and theta brainwave powers. Alpha brainwaves emerge in the awake condition with eyes closed, when the subject is relaxed and processing minimal information. Alpha activity has also been associated with memory recall, lessened discomfort and pain, and reductions in stress and anxiety. Beta brainwaves, on the other hand, signal alertness and attentiveness. A subtype of beta brainwaves, known as low beta or sensorimotor rhythm (SMR) brainwaves, has been associated with mental and emotional disorders, such as depression and insomnia [
14]. Finally, theta brainwaves have been found to usually increase during states of deep relaxation and meditation [
15]. By comparing the measured cortical activity of patients with pruritus with those of the matched healthy subjects, the study sought to determine the effect of itch at the cortical level.
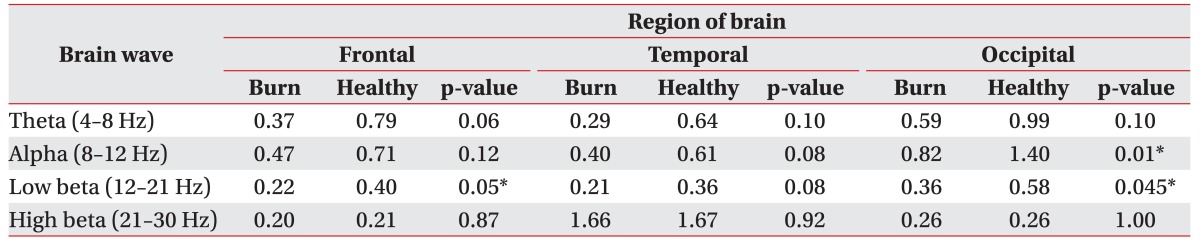
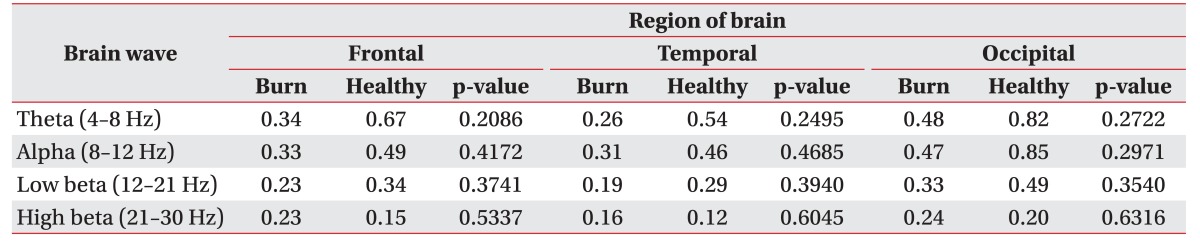
Preliminary results suggest changes in EEG brainwave patterns related to pruritus. Results indicated a decrease in alpha activity in the occipital area and a decrease in low beta activity in the frontal area during the eyes closed condition in burn patients compared to healthy controls. In addition, an overall decrease in theta activity was observed in both the eyes closed and eyes open conditions in burn patients. These brainwave pattern changes are thus evidence of the maladaptive brain reorganization that occurs in the brains of burn patients after pruritus and/or peripheral damage.
Although alpha activity is widely distributed in the cortex, its function is not clear [
16]. However, alpha waves have been found to be associated with meditation and a sense of inner peacefulness created by the release of acetylcholine [
17]. In healthy individuals, an increase in alpha is usually observed during the eyes closed conditions, mainly in the posterior occipital-temporal and parietal regions. Conversely, one study, conducted by Jensen et al. [
8], demonstrated a decrease in alpha activity and an increase in theta activity in the frontal areas in patients with chronic pain after spinal cord lesion in the eyes closed condition. In this study, results also demonstrated a decrease in alpha activity during the eyes closed condition, but, in contrast, in the occipital region. These study results thus replicate the type of alpha activity seen in chronic pain, but in different areas of the brain.
In this study, a trend, though non significant, was observed in the theta bandwidth. Burn patients with itching recorded a lower theta brainwave power compared to the healthy individuals. Theta brainwaves have been correlated to the reward pathway, especially in the frontal components of the pathway as these frontal regions provide executive control over decisions and reward seeking behaviors. The trend of decreased theta activity may be due to the fact that burn patients with itches focus on the itching, thereby causing disruption and distress in their daily activities. Theta waves instigate the release of GABA, a neurotransmitter required to induce relaxation and reduce stress and anxiety, which balances the brain by inhibiting over-excitement [
18]. A decrease in theta waves would thus, in turn, decrease the amount of GABA released. The decreased alpha and theta activity may therefore appear as a result of a lack of relaxation [
12,
14,
19,
20,
21]. However, it is also important to note that one burn participant was taking fluoxetine, an antidepressant. Due to the small sample size, the use of this antidepressant may also account for the decreased trend in the theta oscillations [
22].
Analysis of data from the VAS for sleep and quality of sleep items included in the 5-D pruritus scale indicated that most burn patients recruited in this study reported poor quality of sleep, which may affect their brain reorganization. Specifically, a decrease in theta brainwave activity has been observed in insomniac patients. Many pain studies have suggested changes in alpha and theta activity with disruption to the thalamic-cortical loop [
8,
23,
24]. This disruption, also known as thalamo-cortical dysrhythmia, refers to the abnormal, internally originated low frequency oscillations in the thalamo-cortical network, which disrupts normal connection and communication between these areas. Patients with unsynchronized loops are therefore prone to mental disorders, such as ADD, depression and insomnia. Disruption of the thalamic-cortical loop may thus explain the observed trend of decreased theta brainwaves in burn patients reporting a poor quality of sleep.
Another finding of note is that burn injury patients with pruritus exhibited a significant decrease in low beta brainwaves (12–21 Hz). Low beta brainwaves, also known as SMR brainwaves, allow individuals to feel more present and in the moment. As shown in Egner et al. [
14], training of the low beta brainwave band led to significant attention-enhancing effects in healthy subjects. Low SMR, on the other hand, has been correlated with a lack of focused attention, such as that seen in attention deficit disorder. These low beta brainwaves are inhibited by motion, an issue for burn patients who experience the automatic response to relieve their itching sensation [
11]. Studies have shown that people who lack sufficient beta activity exhibit signs of depression or insomnia, disorders observed in many burn patients as a result of the negative effects of their injury on their lifestyle.
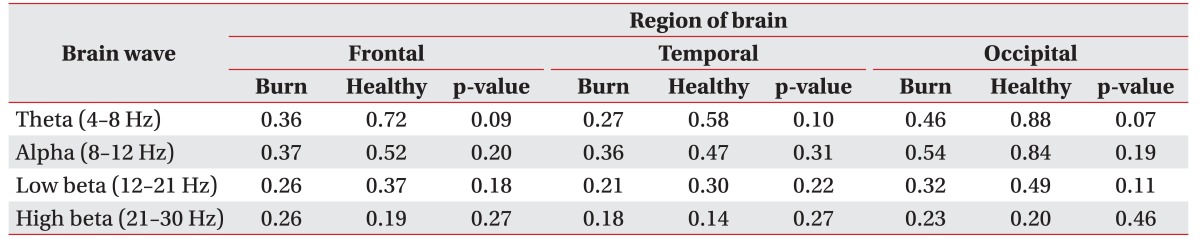
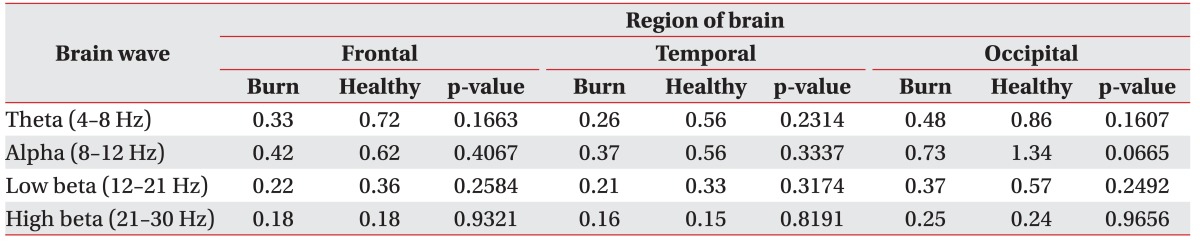
In the eyes open condition, no differences were observed between the two groups, with the exception of a decrease in the overall theta trend. However, results from the eyes open condition were not the study focus, due to the susceptibility to artifacts.
Many other studies have also abstained from including the eyes open condition in their analysis, given that they had not found any significant changes.
These study results provide early findings that itching is processed differently than pain. The reward circuit that emerges after itching, which may create a different network as shown by a different EEG pattern, can also affect the central processing of pruritus. Itching is also associated with conscious and voluntary control motor responses, and the reward process that ensues is associated with the ventral tegmental area (VTA), nucleus accumbens (NAc), and prefrontal cortex (PFC). Another difference identified between pain and itching is the specified pathway in the anterolateral spinothalamic tract fiber by the wide dynamic range (WDR) neurons. These neurons are mechanically insensitive, have very low conduction velocity, and have a high electrical threshold that does not show spontaneous activity [
14]. These differences between the central processing of pain and itching thus demonstrating the need for different forms of treatment to best facilitate efficient recovery of patients with pruritus. Although this study recruited only 4 burn patients, along with their respectively matched healthy controls, significant results were obtained. Further research should still recruit a larger sample size of patients with pruritus post burn injury in order to obtain more accurate results. In addition, the correlation between measures of sleep quality, drowsiness, and emotional states with EEG activity, may also be examined. Furthermore, as the human brain has the capability of reorganization, it is difficult to ascertain whether pruritus occurs as a result of reorganization or peripheral healing [
25]. Therefore, a similar study may be run in the future using burn patients without pruritus as a control group.
The results of this study demonstrate a different EEG pattern in patients with pruritus than that observed in patients with pain. Such results thus add further evidence that pruritus may also cause some maladaptive reorganization and encourage future studies to utilize other forms of recording and brain activity modulation in order to gain insight into the specific mechanism of pruritus at the cortical level. Such research will aim to provide effective treatment plans to improve quality of life outcomes for burn patients experiencing pruritus.