INTRODUCTION
Platinum-analogues, a type of DNA alkylating agent, are important chemotherapeutic drugs which form the basis for treatment for many solid tumor types, including lung, colorectal, gastric, esophageal, head and neck, cervical, bladder, ovarian, and testicular cancer [
1]. However, these agents are known to cause peripheral polyneuropathy as do the taxanes, epothilones, vinca alkaloids, and bortezomib [
2]. Chronic neuro-sensory toxicities caused by these drugs are a major known side effect presenting clinically as distal dysesthesia, pain in glove and stocking forms, decrease in the deep tendon reflex (DTR), and reduced peripheral sensation, ototoxicity, and hearing loss [
3,
4,
5].
While there have been frequent reports of peripheral polyneuropathies or central nervous system involvement resulting from the use of platinum-analogues, reports of cranial neuropathies are rare. Verschraegen et al. [
6] reported visual loss, which occurred after treatment with cisplatin, but this was a symptom reflecting involvement of the cerebral visual cortex and not from peripheral (optic nerve) cranial neuropathy. Some literature have reported that platinum-analogues may induce cranial neuropathy, but most such publications on cranial nerve defects due to platinum therapy are in the form of case reports [
7]. Accordingly, we performed a control group study using the blink reflex on patients who received platinum-analogue chemotherapy and intended to examine whether a cranial neuropathy could be caused by these drugs.
RESULTS
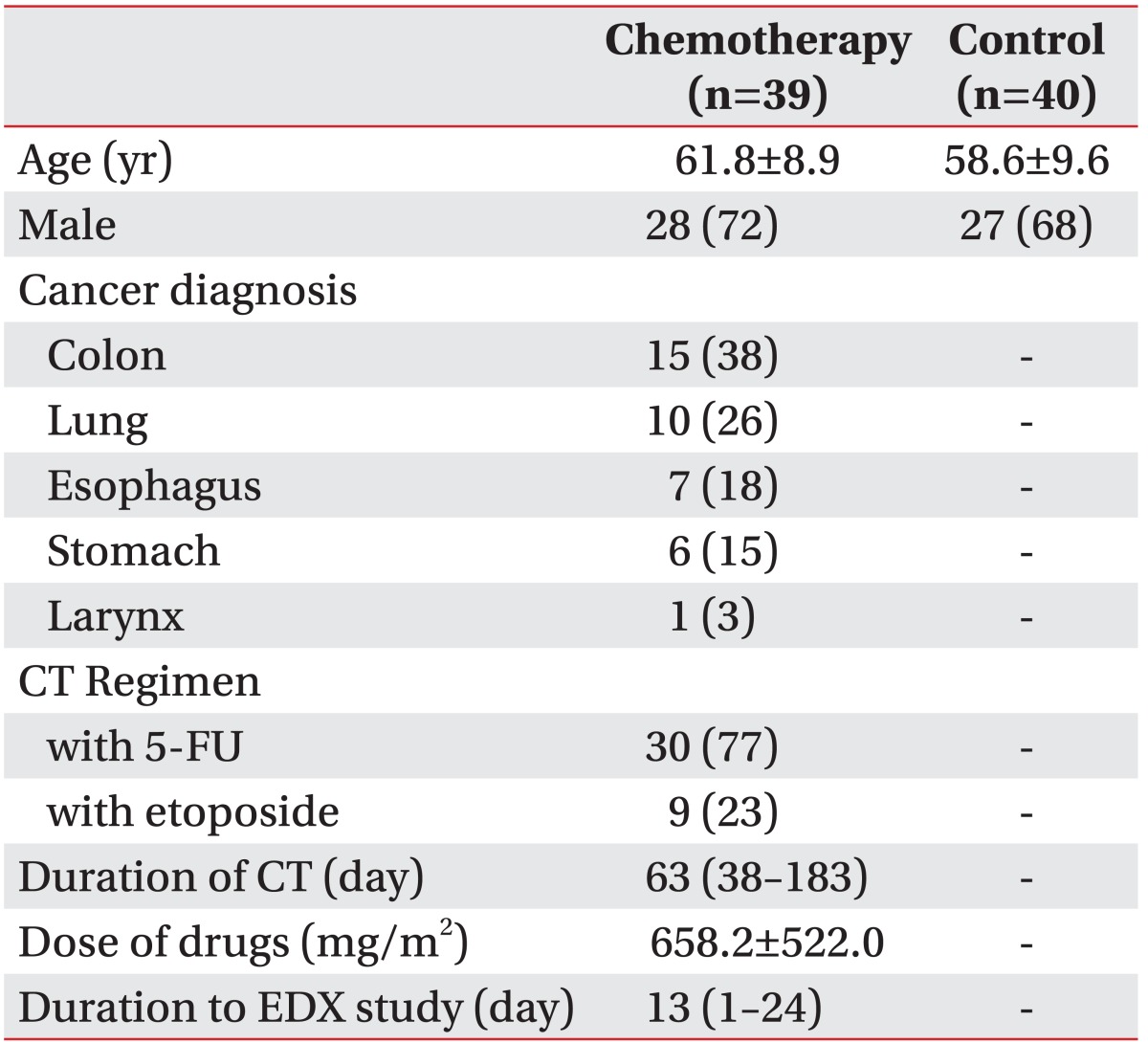
A total of 39 patients (mean age 61.8±8.9 years, 28 males) participated in the present study in the chemotherapy group. The numbers of those whose primary site of cancer was colon and rectum, lung, esophagus, stomach, and larynx were 15, 10, 7, 6, and 1, respectively. The numbers of those who used 5-fluorouracil (5-FU) and etoposide together with platinum agents were 30 and 9, respectively. The control group consisted of 40 patients (mean age 58.6±9.6 years, 27 males) (
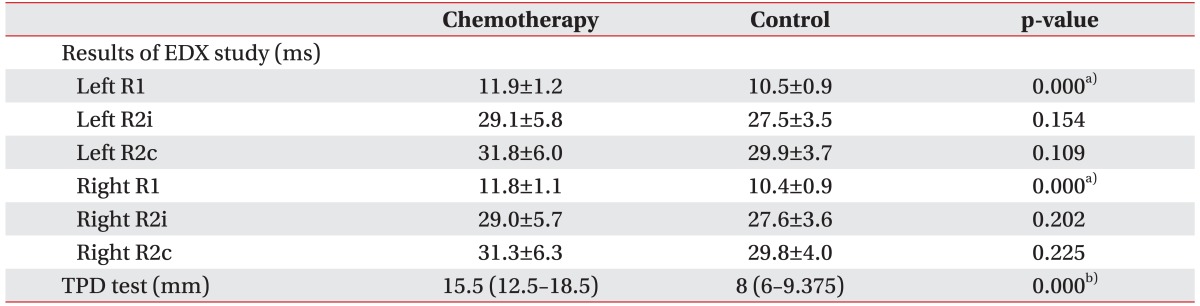
Table 1). In comparing the control and chemotherapy groups, bilateral R1 latencies were significantly prolonged (the left side, 11.9±1.2 ms vs. 10.5±0.9 ms, p<0.001; the right side, 11.8±1.1 ms vs. 10.4±0.9 ms, p<0.001) and the mean distance of two-point discrimination was significantly longer (median [IQR] distance, 15.5 mm [12.5–18.5 mm] vs. 8 mm [6–9.375 mm]; p<0.001) in the chemotherapy group (
Table 2).
The chemotherapy group was divided into two subgroups according to the type of platinum agent: 18 patients with cisplatin and 21 with oxaliplatin. Between the two subgroups, the cumulative dose of the agents significantly differed (432.7±416.7 mg/m
2 vs. 851.4±534.0 mg/m
2; p=0.01), but there was no significant difference when the results of the blink reflex study and two-point discrimination test were compared. In both subgroups, bilateral R1 latencies were significantly prolonged (cisplatin vs. control: the left side, 11.9±1.0 vs. 10.5±0.9, p<0.001; the right side, 11.9±0.9 vs. 10.4±0.9, p<0.001; oxaliplatin vs. control: the left side, 11.9±1.4 vs. 10.5±0.9, p<0.001; the right side, 11.6±1.3 vs. 10.4±0.9, p<0.001) and the mean distance of two-point discrimination was longer (cisplatin vs. control: median [IQR] distance, 14.75 mm [10.375–18.875 mm] vs. 8 mm [6–9.375 mm], p<0.001; oxaliplatin vs. control: 16 mm [12.75–18.5 mm] vs. 8 mm [6–9.375 mm], p<0.001) than in the control group (
Table 3).
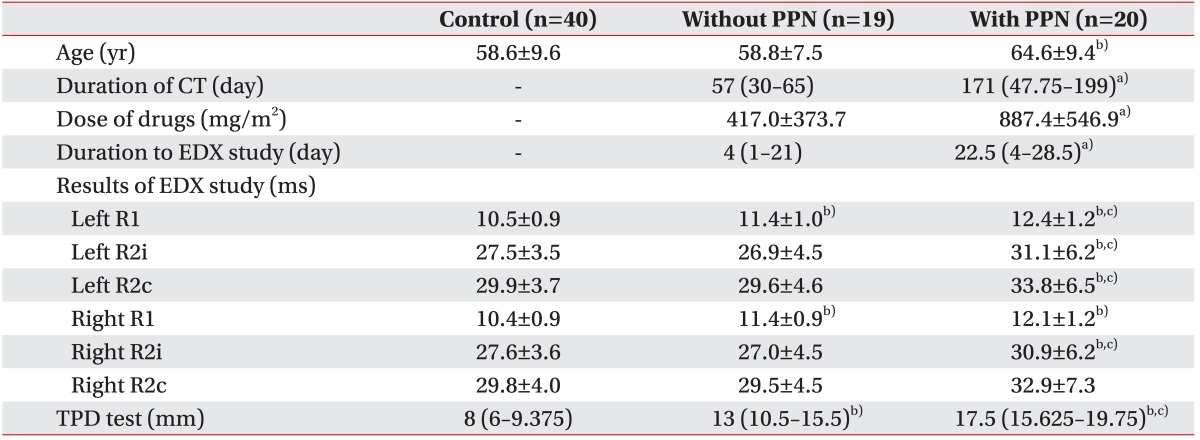
Also, the chemotherapy group was subdivided into two smaller subgroups according to the presence of peripheral polyneuropathy; 20 patients were included in the group with polyneuropathy and 19 in the group without. Those with polyneuropathy were significantly older (64.6±9.4 years vs. 58.8±7.5 years; p=0.04), underwent a longer duration of chemotherapy (median [IQR] duration, 171 days [47.75–199 days] vs. 57 days [30–65 days]; p=0.008), higher cumulative dose of platinum agents (887.4±546.9 mg/m
2 vs. 417.0±373.7 mg/m
2; p=0.003), had a longer period from the completion of chemotherapy to the electrodiagnostic study (median [IQR] period, 22.5 days [4–28.5 days] vs. 4 days [1–21 days]; p=0.008), longer latencies of the left R1 (12.4±1.2 ms vs. 11.4±1.0 ms; p=0.01), R2i (31.1±6.2 ms vs. 26.9±4.5 ms; p=0.02), R2c (33.8±6.5 ms vs. 29.6±4.6 ms; p=0.02), and the right R2i (30.9±6.2 ms vs. 27.0±4.5 ms; p=0.03) responses, and greater distance of two-point discrimination (median [IQR] distance, 17.5 mm [15.625–19.75 mm] vs. 13 mm [10.5–15.5 mm]; p=0.008). In the subgroup without the polyneuropathy, bilateral R1 latencies were significantly prolonged (the left side, 11.4±1.0 vs. 10.5±0.9, p=0.006; the right side, 11.4±0.9 vs. 10.4±0.9, p<0.001), and the mean distance of two-point discrimination was longer (median [IQR] distance, 13 mm [10.5–15.5 mm] vs. 8 mm [6–9.375 mm]; p<0.001) than in the control group. In the subgroup with the polyneuropathy, bilateral R1 (the left side, 12.4±1.2 vs. 10.5±0.9, p<0.001; the right side, 12.1±1.2 vs. 10.4±0.9, p<0.001), R2i (the left side, 31.1±6.2 vs. 27.5±3.5, p=0.01; the right side, 30.9±6.2 vs. 27.6±3.6, p=0.03), the left R2c (33.8±6.5 vs. 29.9±3.7, p=0.01) latencies, and mean distance of two-point discrimination (median [IQR] distance, 17.5 mm [15.625–19.75 mm] vs. 8 mm [6–9.375 mm]; p<0.001) showed significant differences compared to the control group (
Table 4).
DISCUSSION
Platinum-derivatives are important chemotherapeutic agents that form the mainstay for treatment of lung, colorectal, gastric, head and neck, cervical, esophageal, bladder, ovarian, and testicular cancer in combination with radiation therapy and/or other chemotherapeutic agents [
1]. Since 1978, when cisplatin, the first-generation platinum agent, was approved for the treatment of ovarian and testicular cancer [
9], development of other platinum analogues has developed into forms associated with less toxicity and greater treatment efficacy. Carboplatin is a second-line platinum chemotherapeutic agent used for the treatment of lung cancer in combination with gemcitabine or paclitaxel [
10], and the third-generation oxaliplatin is utilized for treating colorectal cancer together with 5-FU and leucovorin [
11]. Neurotoxicity of platinum agents and the consequent peripheral polyneuropathy remains a major non-hematological dose-limiting side effect. Although research for the prevention or minimization of polyneuropathy (e.g., glutathione and vitamin E) is continuing, there has yet been no development of effective treatment or prevention [
12].
Cranial neuropathy resulting from anti-cancer treatment has been reported to be associated with chemotherapeutic agents such as vincristine, cyclosporine, and cytarabine [
7]. Toker et al. [
13] reported diplopia caused by abducens nerve paralysis following chemotherapy with vincristine for three weeks in a 28-year-old non-Hodgkin's lymphoma patient. Vogel and Horoupian [
14] reported dysarthria and ophthalmoplegia which occurred after high-dose cytarabine treatment in a 43-year-old patient with acute promyelocytic leukemia and was found to have involvement of the brainstem and spinal motor neuron. Verschraegen et al. [
6] reported a case of blindness, which occurred after cisplatin treatment, but this was determined to be due to involvement of the cerebral visual cortex rather than by a cranial neuropathy. Research on peripheral polyneuropathy or CNS side effects resulting from chemotherapy have been frequently reported. However, the literature on platinum chemotherapy- associated cranial neuropathy are limited to few, isolated case reports. Accordingly, the authors planned this control group study in order to evaluate whether cranial neuropathies could be caused by platinum agents. In the present study, the chemotherapy group showed delay of the bilateral R1 latencies in the blink reflex study and significant hypoesthesia in the two-point discrimination test compared to the control group (
Table 2). These findings support the occurrence of subclinical cranial neuropathy caused by platinum-analogues chemotherapy.
In the present study, the chemotherapy group demonstrated a delay in the blink reflex including with bilateral R1, R2i, and R2c latencies compared to the control group, although statistical significance was found only in the bilateral R1 latencies (
Table 2). In peripheral polyneuropathy caused by platinum agents, the dorsal root ganglia (DRG), which lacks an effective blood-brain barrier (BBB), are considered to be the primary targets of the drugs, which cause apoptosis of sensory neurons [
15,
16]. Similarly, in our patients, extra-axial lesions, including the sensory trigeminal roots, may be exposed to chemically- induced damage and, consequently, all the R1, R2i, and R2c latencies would probably be prolonged. In addition, Guney et al. [
17] reported blink reflex alterations in diabetic patients, concluding that in the patients with extra-axial lesions, R1 is more sensitive than R2. Furthermore, the R1 response is known to be mainly conducted by exteroceptive, medium-thick myelinated A-beta (Aβ) fibers, whereas the R2 response is predominantly conveyed by nociceptive, thin myelinated A-delta (Aδ) fibers [
18]. In the previous studies of cisplatin neurotoxicity, large myelinated sensory nerve fibers were more vulnerable to chemotherapy and the decrement of mediumand large-sized neuron was observed in the patients' peripheral nerves [
19,
20,
21]. We hypothesized that these results may be helpful in explaining the more profound change of R1 than R2 in our patients.
Because our patients did not show any symptoms or signs suggestive of a cranial neuropathy, we performed clinical evaluation with the two-point discrimination test as well as with electrodiagnostic studies. We found that the chemotherapy group showed significant hypoesthesia compared to the control group (
Table 2). Thus, the cranial nerve involvement by platinum agents led to overt clinical manifestations, in addition to the elctrophysiological findings. In addition, given that mechanical stimulation including two-point discrimination sense was conducted through the Aβ fibers, it could be interpreted as a consistent result with the more profound delay of R1 than R2 latencies in the chemotherapy group.
Neurotoxicity caused by platinum agents shows a cumulative dose effect. In the case of cisplatin, signs of neurotoxicity, such as a decrease or loss of DTR, begin to appear after a cumulative dose of greater than 300 mg/m
2, and, therefore, most patients who finish chemotherapy with the usual dose (300–450 mg/m
2) experience sensory neuropathy symptoms. In addition, after treatment with 750–850 mg/m
2 of oxaliplatin, 82%–93% of patients experience neuropathic symptoms. It is known that the severity of neuropathy worsens as the cumulative dose of the drugs increase [
22]. In subgroup analysis of the present study, the patients with polyneuropathy showed bilateral R1 and R2 delay compared to the control group, whereas those without the polyneuropathy showed only R1 delay. In addition, those with polyneuropathy showed significant delay of bilateral R1 and R2 latencies and had longer duration of chemotherapy and higher cumulative dose of platinum agents compared to those without the polyneuropathy (
Table 4). It is likely that the platinum agents were neurotoxic to the patient's PNS, including the cranial nerves. Therefore, involvement of the cranial nerves may be more profound in patients with peripheral polyneuropathy and may be dependent on the cumulative dose of the drugs. On the other hand, the finding of the R1 delay of both the patients with and without the polyneuropathy may indicate that that R1 is more sensitive in detecting subclinical cranial neuropathy than is R2. In general, most peripheral polyneuropathies affect the lower limb nerves and sensory fibers before the upper limb nerves and motor fibers, respectively. However, that platinum-based chemotherapy may preferentially affect limb over cranial nerves has yet to be definitively defined. Similarly, Elkholy et al. [
23] and Resende et al. [
24] also reported blink reflex alterations not accompanied by peripheral polyneuropathy in patients with diabetes and end-stage renal disease.
The present study has some limitations. A major limitation was that the study design was cross-sectional and comparisons were made between patients and healthy controls. For future studies, comparison of the blink reflex before-and-after chemotherapy or, alternatively, between symptomatic and asymptomatic patients may be more ideal. However, most of the patients who underwent the chemotherapy were asymptomatic, even those with symptoms of peripheral polyneuropathy. Therefore, further study with before-and-after evaluations may reach a more valid conclusion. Secondly, the present study failed to obtain the homogeneity of chemotherapy regimen in the chemotherapy group. The most ideal study design would be to include patients who have received single agent chemotherapy participate in the chemotherapy group, but have most other patients on anti-cancer treatment receive combination chemotherapy, except for those who undergo chemotherapy with adjuvant treatment. In addition, patients who receive adjuvant chemotherapy receive lower cumulative doses of chemotherapeutic agents, and therefore, may not be appropriate as subjects of the study evaluating neurotoxicity. Accordingly, despite the use of combination chemotherapy, only patients who additionally used 5-FU or etoposide, which were known to have little neurotoxicity [
22], were included as the subjects (in order to minimize the confounding effect of other chemotherapeutic agents). Thirdly, the chemotherapy group was divided into two subgroups according to whether they had peripheral polyneuropathy, and their cumulative dose of platinum agents was compared (
Table 4). However, the usual dose of cisplatin (300–450 mg/m
2) and oxaliplatin (≥700 mg/m
2) were greatly different from each other. For thease reasons, comparing their relative dose effect may not be appropriate. For the same reason, significant dose differences between the subgroups of cisplatin and oxaliplatin may not be adequately interpreted at face value (
Table 3). Further research with larger numbers of patients comparing the dose for each chemotherapeutic agents (i.e., cisplatin or oxaliplatin) should be performed. Finally, the present study did not perform long-term follow-up observation of cranial neuropathy. Neuropathy caused by platinum agents may continue or worsen in the few weeks following completion of the chemotherapy, a phenomenon coined the coasting phenomenon [
25]. Because we conducted electrodiagnostic studies and the two-point discrimination test within one month after finishing the chemotherapy, this strategy was effective for recruiting patients and preventing their loss to follow-up. Nonetheless, it remained difficult to judge whether evaluations were made after the cranial neuropathy sufficiently appeared. Accordingly, further research, including long-term follow-up observation by other known objective methods such as facial motor NCS, trigeminal somatosensory evoked potential, visual evoked potential, auditory evoked potential, vestibular evoked myogenic potential and masseter inhibitory reflex in company with blink reflex, is now indicated.
In the present study, comparison of the blink reflex study and two-point discrimination test demonstrated the likelihood of subclinical cranial neuropathy following platinum-analogues chemotherapy. Cranial neuropathy caused by the platinum agents was more profound in patients with peripheral polyneuropathy and may be dependent upon the cumulative dose of the drugs. The blink reflex presumably is of value in detecting subclinical cranial neuropathy in patients with platinum-analogues chemotherapy.