- Search
| Ann Rehabil Med > Volume 42(6); 2018 > Article |
|
Abstract
Objective
To elucidate the effect of a 12-week horizontal vibration exercise (HVE) in chronic low back pain (CLBP) patients as compared to vertical vibration exercise (VVE).
Methods
Twenty-eight CLBP patients were randomly assigned to either the HVE or VVE group. All participants performed the exercise for 30 minutes each day, three times a week, for a total of 12 weeks. Altered pain and functional ability were evaluated using the visual analog scale (VAS) and Oswestry Disability Index (ODI), respectively. Changes in lumbar muscle strength, transverse abdominis (TrA) and multifidus muscle thicknesses, and standing balance were measured using an isokinetic dynamometer, ultrasonography, and balance parameters, respectively. These assessments were evaluated prior to treatment, 6 weeks and 12 weeks after the first treatment, and 4 weeks after the end of treatment (that is, 16 weeks after the first treatment).
Results
According to the repeated-measures analysis of variance, there were significant improvements with time on VAS, ODI, standing balance score, lumbar flexor, and extensor muscle strength (all p<0.001 in both groups) without any significant changes in TrA (p=0.153 in HVE, p=0.561 in VVE group) or multifidus (p=0.737 in HVE, p=0.380 in VVE group) muscle thickness. Further, there were no significant differences between groups according to time in any of the assessments. No adverse events were noticed during treatment in either group.
Low back pain (LBP) is one of the most common types of pain, occurring at least once in a lifetime in more than 80% of the total population [1]. In most cases, LBP usually resolves within 8 to 12 weeks; however, it develops into chronic low back pain (CLBP) in about 15% of acute cases [2,3]. Several studies have suggested risk factors of LBP, including psychological stress, structural defects of the spine, and biomechanical and genetic factors [4]. However, in 85% of cases of CLBP, no specific cause can be determined from imaging or biomechanical analysis, and this type of LBP is classified as non-specific CLBP [3]. LBP distorts normal signals entering the motor and sensory organs and prevents the maintenance of normal balance [5]. The chronicity of such pain leads to immobilization following disuse atrophy of the muscles, especially when the relevant muscles lose their stabilization ability [6].
According to most clinical guidelines, exercise training is known to effectively reduce pain and improve back function in CLBP [7]. However, some patients cannot participate in conventional vigorous exercise training due to prior illness [8]. Whole body vibration (WBV) exercise has been used to train musculoskeletal systems at homes and rehabilitation clinics. Because of its simplicity and ease, WBV is appropriate for the elderly as well as patients who cannot participate in traditional exercise [9]. The vibrations generated by the vibrating platform transmitted through the body stimulate all sensory receptors within the epidermis, dermis, joint capsules, and muscle spindles [10]. The stretch reflex is activated by changes in the length of the muscle spindles, which enhances the frequency of motor evoked potentials and in turn improves neuromuscular performance [10,11]. WBV is useful for improving pain relief as well as restoring balance, muscle strength, and quality of life in patients with non-specific CLBP [11]. Rittweger et al. [12] reported improved pain and function in patients with CLBP after a 12-week WBV therapy. These positive effects are the result of improved proprioception and improved muscular coordination of the lumbo-pelvic region [12].
Based on these positive effects, WBV has been increasingly used for relief from musculoskeletal pain, and may complement standard physical rehabilitation treatment [7,13]. Several reports have suggested that WBV exercise might be effective for CLBP, but most of them investigated vertical vibration by rotational motion [11,12,14]. Although WBV is effective and safe, a few studies have reported that vertical vibration might be a harmful stimulus to the body. Several case reports have described dislocation of the intraocular lens or vitreous hemorrhage of the eye following vertical WBV [15,16]. Repetitive high dose vertical vibration can induce fatigue injury in fragile bones and may potentially lead to damage to aged cartilage tissue [17]. Recently, a WBV medical device (EXXTREAM 1000; AMH International Ltd., Incheon, Korea) was developed to convert rotational motion into horizontal vibration, and is expected to reduce the harmful body pressure as compared to vertical vibration. Several studies have reported improved balance and motor function in horizontal WBV [18,19]. However, there has been no study comparing the effect of HVE to that of conventional VVE on CLBP.
Thus, the purpose of this study was to elucidate the effects on CLBP patients of a 12-week horizontal axis vibration exercise as compared to a vertical axis vibration exercise for pain, functional disability, standing balance, lumbar muscle strength, and abdominal and paraspinal muscle thicknesses.
Twenty-eight patients who visited or were referred to the rehabilitation center of Dongguk University Ilsan Hospital for LBP were recruited. The inclusion criteria were: CLBP without any major neurological deficit and a history of at least 6 months. We investigated the past medical history and radiologic study of each patient so as to exclude the specific lesions which could either be worsened by vibration exercise or influence the evaluation parameters. The exclusion criteria were: (1) severe cardiovascular or central nervous system diseases, such as stroke or Parkinson disease; (2) vestibular dysfunction or peripheral nervous disease; (3) history of vertebral diseases, such as severe osteoporosis (T-scoreŌēż-2.5), spinal surgery (within the past 6 months), compression fractures of the axial bone (>40% reduction of any vertebral height), or spondylolisthesis (>50% slip); (4) change of any treatments that could influence pain, muscle strength, or balance within the past 6 months; (5) scheduled spinal intervention within 3 months prior to enrollment or any other therapy for LBP during the trial; (6) patients who found it difficult to undergo knee flexion posture. The sample size was calculated by G*Power 3.1.9.2 (effect size f=0.25, a=0.05, power=0.80, number of groups=2, number of measurements=4) for repeatedmeasures analysis of variance (ANOVA). The total sample size was 24. A total of 28 participants were recruited in consideration of the dropout rate of 15%.
According to the type of exercise, subjects were randomly assigned to the horizontal vibration exercise (HVE) or vertical vibration exercise (VVE) groups using a table of random numbers; they were assigned to different training sessions for blinded intervention. In addition, we provided no information involving the differences in the treatment methods. The vibration exercise was conducted by expert physical therapists, and the assessment was performed by medical doctors and researchers who were blinded to group allocation. All participants performed the exercise program for 30 minutes a day, three times a week, for a total 12 weeks. The program consisted of 5 minutes of warming-up exercise followed by 20 minutes of WBV exerciseŌĆö10 minutes of antero-posterior (AP) direction +10 minutes of medio-lateral (ML) direction after postural change, and then 5 minutes of cool-down exercise. The subjects were asked to stand barefoot on the platforms in order to prevent damping of the vibrations. Both angles of the knee flexion were maintained around 60┬░ so as to reduce the transmission of the vibrations to the head and spine (Fig. 1). Subjects held the handle of the device to maintain their balance during the vibration exercise.
In HVE, the trials were performed under horizontal vibration in the AP plane with a frequency of 3ŌĆō5 Hz and an amplitude of 1ŌĆō48 mm in the sagittal axis. The intensities of frequency and amplitude ranged from 5 to 15 levels and gradually increased to the maximum tolerance limit. In VVE, the subjects used conventional vertical vibration similar to HVE, except that the vibration was in the vertical axis with a frequency range of 28ŌĆō34 Hz and an amplitude of 2.5ŌĆō5 mm. In order to blind the subjects, the same treatment protocol as in the HVE group was applied for the same duration in the VVE group. The intensity of treatment was controlled by gradually increasing the frequency to the maximum tolerance limit, and was adjusted for each treatment session.
The participants were assessed prior to exercise (preintervention, t0), 6 weeks after the first exercise (mid-intervention, t1), 12 weeks after the first exercise (post-intervention, t2), and 4 weeks from the last exercise session (follow-up, t3). The evaluation parameters included visual analog scale (VAS), Oswestry Disability Index (ODI), standing balance control score, lumbar muscle strength, and abdominal and paraspinal muscle thicknesses.
The primary outcome measures in this study were VAS of back pain and ODI. The secondary outcome measures were lumbar muscle strength, abdominal and paraspinal muscle thickness, and the standing balance control score. Lumbar muscle strength was measured with an isokinetic dynamometer (Model 900-240; Biodex Medical Systems, Shirley, NY, USA). The maximal and average voluntary lumbar extension and flexion strength were obtained as well [20]. Abdominal and paraspinal muscle thicknesses were measured using ultrasonography. For the abdominal muscle, the thickness of the transverse abdominis (TrA) was measured at the junction of the right iliac crest with the axillary line, while for the paraspinal muscle, the thickness of the L4ŌĆō5 level multifidus muscle was measured. The spine corresponding to the level of the iliac crest was regarded as a reference point for L4 lumbar vertebrae. Based on this criterion, the spinal processes were marked on the skin with marker from L1 to S1. The ultrasound transducer was positioned longitudinally on the line connecting the spinal processes and was moved laterally so as to obtain a parasagittal image of the multifidus [21]. The thickness of the multifidus muscle was used to measure the distance from the most superficial plane of the L4ŌĆō5 facet joint to the plane between the muscle and the subcutaneous tissue [22]. For all measurement variables, the average of the two values obtained from the images on both sides was used for analysis.
Standing balance control scores were determined using a Biodex Stability System (Biodex Medical Systems). Patients were instructed to stand barefoot on the platform with a slight knee flexion. After initialization of the center of pressure by quiet standing, three stability trials were performed in 30 seconds by altering the platform. The weight shift was tracked by a green line on a monitor divided into four quadrants [23]. The device measured and analyzed the ML and AP indices as mean┬▒standard deviation. The higher the index scores, the greater the instability; the ML index represents the stability of the coronal plane while the AP index represents the stability of the sagittal plane [24].
Statistical analysis was performed using statistical software, SPSS version 20.0 (IBM SPSS, Armonk, NY, USA). Statistical significance was determined at p-values less than 0.05. Demographic and baseline parameters were analyzed by independent t-tests or chi-squared tests. The sphericity of the data was verified with a Mauchly test. The Greenhouse-Geisser correction was applied when the assumption of sphericity was violated. In order to investigate and compare the changes of each outcome at each period (t0, t1, t2, and t3) the variables were analyzed with repeated measures ANOVA using time and group as factors. In the presence of significance, Bonferroniadjusted pairwise comparisons were used.
This study was approved by the Institutional Review Board of the Dongguk University Ilsan Hospital (No. 2013-112), and written informed consent was obtained from every participant prior to study initiation.
A total of 28 participants completed all of the exercise and assessment sessions. No study-related adverse events were noted during treatment in either of the groups. The mean ages were 55.1┬▒11.2 years in the HVE group and 53.7┬▒12.1 years in the VVE group. No significant differences in were detected in the baseline characteristics of both groups (Table 1).
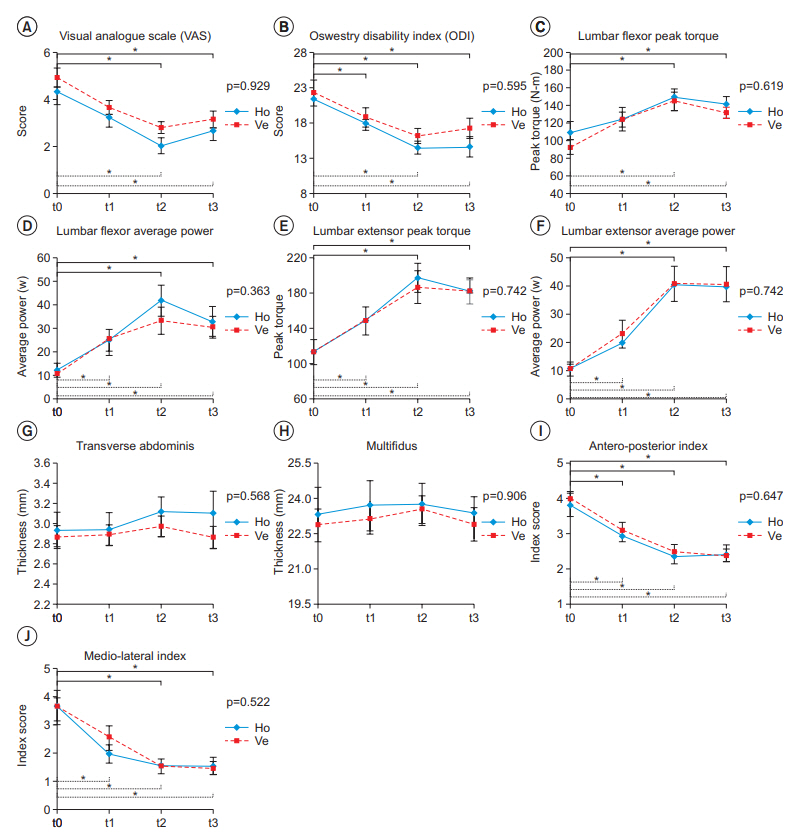
VAS scores for pain were significantly reduced over time in both the HVE and VVE groups (p<0.001). In both groups, a significant decrease in VAS score was detected from 12 weeks (t0 vs. t2, p=0.001 in HVE; t0 vs. t2, p=0.002 in VVE) that persisted until 4 weeks after the last exercise session, as compared to baseline (t0 vs. t3, p=0.04 in HVE; t0 vs. t3, p=0.005 in VVE). However, the differences between groups according to time were not significant (p=0.929) (Table 2, Fig. 2A).
ODI was significantly reduced over time in both groups (p<0.001). In the HVE group, a significant decrease in ODI occurred at 6 weeks of treatment (t0 vs. t1, p=0.003) that persisted until 4 weeks after the last exercise session (t0 vs. t3, p=0.00). In the VVE group, a significant decrease was found from 12 weeks (t0 vs. t2, p=0.011), which persisted until 4 weeks after the last exercise session (t0 vs. t3, p=0.003). However, there was no significant difference between groups according to time (p=0.595) (Table 2, Fig. 2B).
In terms of peak torque and average power, all measurements of lumbar trunk muscle strength significantly increased over time in both groups (p<0.001). Compared with the baseline, a significant increase was observed at 12 weeks (t0 vs. t2, p<0.01 in all measurements of strength) and it persisted until 4 weeks after the last exercise session (t0 vs. t3, p<0.01 in all measurements of strength) in the HVE group. In the VVE group, a significant increase occurred from 6 weeks of treatment (t0 vs. t1, p<0.01 in all measurements of strength) and it persisted until 4 weeks after the end of treatment (t0 vs. t3, p<0.01 in all measurements of strength). However, there was no significant group difference with time in all assessments (Table 2, Fig. 2C-2F).
There were no significant changes in either TrA or multifidus muscle thickness over time in the HVE group (p=0.153 in TrA, p=0.737 in multifidus) or VVE group (p=0.561 in TrA, p=0.380 in multifidus), although muscle strength increased significantly. There were no significant differences in the thickness of either muscle between the two groups (p=0.568 in TrA, p=0.906 in multifidus) (Table 2, Fig. 2G, 2H).
The standing balance control scores improved significantly over time in both groups (p<0.001). The balance score of the AP plane showed a significant difference at 6 weeks in both groups (t0 vs. t1, p=0.017 in HVE, p=0.00 in VVE). The balance score of the ML plane showed a significant difference at 6 weeks in the VVE group (t0 vs. t1, p=0.008) and 12 weeks in the HVE group (t0 vs. t2, p=0.005), both of which persisted until 4 weeks after the last exercise session. However, there were no significant differences between the groups over time in either of the AP and ML planes (p=0.647 in AP, p=0.522 in ML) (Table 2, Fig. 2I, 2J).
Traditional WBV involves a vertical vibration induced by the rotational motion of a motor. EXXTREAM 1000 is a newly-developed medical device which converts rotational motion into horizontal vibrations. Studies investigating the effects and mechanisms of horizontal vibrations are rare, and there is no universal consensus. Lee reported a significant improvement in motor function and balance using the horizontal WBV with a conventional rehabilitation group of chronic stroke survivors, but not in the control group, which received conventional rehabilitation only [18]. Shim et al. [19] reported that elderly people who underwent horizontal WBV showed significant improvements in balance and fear of falling. Bagheri et al. [10] compared the acute effects of the three types of vibration of horizontal, vertical, and mixed on muscle performance. There were no significant differences between the three types of stimulation for jump force and jump rate of force development in 12 healthy subjects. However, no studies to date have compared HVE with VVE in CLBP patients.
There are several theories of pain reduction in WBV. Increased pain sensitivity or reduced pain threshold is known to be a major pain mechanism in CLBP [12]. Non-noxious stimulus induced by vibration reduces the number of activated spinothalamic tract neurons as well as the synchronicity of the active volley reaching the cerebral hemisphere. This effect reduces pain and elevates the pain threshold [25]. In addition, based on the gate control theory, an interference stimulus such as vibration arrives in the brain prior to the pain stimulus, which may also reduce pain perception [26]. Humans cannot completely distinguish pain from other sensory stimuli being administered concurrently, and tend to disregard unpleasant sensations [25]. Thus, only vibration sensations that reach the brain first can reduce painful sensations.
WBV exercise has been shown to improve muscle performance and strength [27,28]. Several studies have reported an increase in the endurances of abdominal and multifidus muscles in CLBP patients following WBV exercise [29,30]. The activation of a larger fraction of the motor neuron pool and the recruitment of previously inactive motor units into contraction results in more efficient use of the force production potential [29,30]; this is considered to be the major effect of vibration exercise. In our study, muscle strength and performance improved without muscle hypertrophy. It is possible that horizontal vibration in the AP or ML direction may evoke the tonic vibration reflex (TVR) by changing the length of the muscle spindles, consequently increasing muscle activity and the efficiencies of the lumbar flexor and extensor [31]. It has also been suggested that WBV exercise restores proprioception and facilitates the improvement in muscle coordination of the lumbar area muscle by activating the mechanoreceptors in the lumbo-pelvic region [32].
The main difference between HVE and VVE is the vibrational direction. In a sense, a horizontal perturbation mobilizes the body in the AP or ML direction, similar to the weight shifting exercise [18]. The weight shifting exercise facilitates the contraction of muscles in the lower extremities as well as core muscles, and has also been reported to improve balance control [18,33]. In addition, the activations and strengthening of abdominal and back extensor muscles reduce pain in CLBP patients [12]. Therefore, HVE was expected to be more effective than VVE in reducing pain, improving disability, and the balance of posture, because core muscles are strengthened by maintaining posture against perturbation. However, there were no significant differences between the HVE and VVE groups in this study, for several reasons. First, we only compared the short-term outcomes of both WBV groups because of the short intervention period. Vibration waves stimulated the muscle spindle, which activated a larger motor neuron pool and recruited previously inactive motor units into contraction, which resulted in a more efficient use of the force generated in the involved muscle groups [27,30]. This effect was enhanced by the synchronization of motor neuron activity, an increased discharge of the neutral drive that led to an improvement in neural-motor control during spontaneous muscle contraction [30]. This processes is called neural adaptation, which explains the increase in early muscle strength and improved muscle tension by resistance exercise [34]. It has been reported that neural adaptation begins within a few months, whereas structural changes such as muscle hypertrophy are further delayed [27]. In other words, changes in the core muscle thickness may not appear in such a short intervention period. This hypothesis requires validation in a long-term intervention study of the effects of HVE.
Second, predictable perturbations may have affected the outcome. Unexpected perturbation training improves oneŌĆÖs ability to maintain standing balance control [35]. The central nervous system increases the reliance on the feedback corrective mechanism based on trial and error of the perturbation stimuli [35]. Further, unpredictable random vibration led to greater activation of the supplementary motor area (SMA) and prefrontal areas as compared to regular vibration [36,37]. SMA is known to play an important role in the generation and regulation of complex movements [36]. We provided a horizontal WBV with fixed amplitude and frequency, and predictable perturbation training might result in less-than-expected results. Thus, follow-up studies are needed to provide unpredictable perturbation via changes in frequency, amplitude, and direction.
This study has a few limitations. First, it was difficult to investigate the long-term effect and the effect of lasting training of HVE on CLBP because of the short intervention and follow-up periods. Second, we failed to investigate the effect of leg muscle strength on standing balance. Third, the research group in this study was not compared with a conventional core exercise group, because HVE has been shown to be as effective as conventional core exercise on CLBP patients, and because the purpose of this article was to compare the effects of HVE and VVE on CLBP patients. However, for the clinical application of HVE, it is necessary to compare the effects of HVE with conventional therapy. Further studies are warranted to explore this issue, and these would require a risk-benefit analysis of HVE.
To the best of our knowledge, this is the first study to compare the comprehensive effects following the application of horizontal or vertical vibrations in CLBP patients. According to this study, both HVE and VVE are effective intervention methods for reducing pain and improving functional disability, standing balance, and lumbar muscle strength in patients with CLBP. However, no statistically significant hypertrophy of the TrA or multifidus muscle was detected over time. In addition, there was no significant temporal difference between the groups in any of the assessments. Based on these findings, we conclude that HVE is as effective as VVE for CLBP patients.
Fig.┬Ā1.
A participant stands on the vibrating platform of horizontal whole-body vibration (EXXTREAM 1000) with flexions of both knees.

Fig.┬Ā2.
Comparison between horizontal vibration exercise (HVE) and vertical vibration exercise (VVE) groups. (A) Visual analog scale, (B) Oswestry disability index, (C) lumbar flexor peak torque, (D) lumbar flexor average power, (E) lumbar extensor peak torque, (F) lumbar extensor average power, (G) transverse abdominis, (H) multifidus, (I) anteroposterior index, and (J) medio-lateral index. All variables except for muscle thickness were significantly changed according to time in each group; however, no variables with significant time-group interactions between the groups existed. All results were obtained using repeated-measures of ANOVA. Ho, horizontal vibration group; Ve, vertical vibration group; t0, assessment at baseline; t1, assessment at 6 weeks; t2, assessment at 12 weeks; t3, assessment performed after 4 weeks from the end of intervention. *p<0.05, a significant difference according to time in each group (solid line, difference in horizontal vibration exercise group; dotted line, difference in vertical vibration exercise group.
Table┬Ā1.
Baseline demographic and general characteristics
| Variable | Ho group (n=14) | Ve group (n=14) | p-value |
|---|---|---|---|
| Sex (male:female) | 4:10 | 3:11 | 1.00 |
| Age (yr) | 55.1┬▒11.2 | 53.7┬▒12.1 | 0.76 |
| Height (cm) | 162.1┬▒6.7 | 160.1┬▒6.9 | 0.46 |
| Weight (kg) | 56.1┬▒9.3 | 56.6┬▒7.0 | 0.86 |
Table┬Ā2.
Changes in clinical outcomes during and following the training of HVE or VVE
| Outcome measure | Group | t0 | t1 | t2 | t3 |
The change by time |
Between group difference for the change by time (p-value) | t0 vs t1 | t0 vs t2 | t0 vs t3 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ho | Ve | ||||||||||
| VAS | Ho | 4.3┬▒1.5 | 3.2┬▒1.1 | 2.0┬▒0.9 | 2.6┬▒1.0 | <0.001 | <0.001 | 0.929 | 0.059 | 0.001* | 0.040* |
| Ve | 4.9┬▒1.9 | 3.6┬▒1.6 | 2.8┬▒1.3 | 3.1┬▒1.5 | 0.135 | 0.002* | 0.005* | ||||
| ODI | Ho | 21.57┬▒4.11 | 18.07┬▒3.50 | 14.57┬▒3.67 | 14.71┬▒5.47 | <0.001 | <0.001 | 0.595 | 0.003* | 0.000* | 0.000* |
| Ve | 22.36┬▒6.76 | 18.86┬▒5.04 | 16.21┬▒4.02 | 17.43┬▒5.42 | 0.158 | 0.011* | 0.003* | ||||
| Muscle strength | |||||||||||
| ŌĆāLFPT (N┬Ęm) | Ho | 109.05┬▒46.67 | 124.44┬▒48.87 | 150.24┬▒32.54 | 140.98┬▒38.74 | <0.001 | <0.001 | 0.619 | 0.758 | 0.001* | 0.012* |
| Ve | 92.51┬▒30.23 | 124.26┬▒32.87 | 144.54┬▒38.06 | 131.74┬▒22.30 | 0.003* | 0.001* | 0.001* | ||||
| ŌĆāLEPT (N┬Ęm) | Ho | 112.89┬▒52.45 | 147.91┬▒54.60 | 197.34┬▒63.79 | 182.64┬▒54.32 | <0.001 | <0.001 | 0.742 | 0.069 | 0.000* | 0.001* |
| Ve | 113.04┬▒82.72 | 148.91┬▒57.60 | 186.36┬▒70.61 | 181.63┬▒52.69 | 0.002* | 0.000* | 0.000* | ||||
| ŌĆāLFAP (W) | Ho | 12.67┬▒9.55 | 24.26┬▒20.76 | 41.78┬▒24.80 | 33.11┬▒23.50 | <0.001 | <0.001 | 0.363 | 0.062 | 0.000* | 0.005* |
| Ve | 10.32┬▒7.30 | 24.30┬▒17.48 | 32.62┬▒22.48 | 29.89┬▒17.97 | 0.017* | 0.007* | 0.003* | ||||
| ŌĆāLEAP (W) | Ho | 10.58┬▒6.99 | 20.16┬▒14.56 | 40.38┬▒24.79 | 39.59┬▒27.26 | <0.001 | <0.001 | 0.878 | 0.067 | 0.002* | 0.003* |
| Ve | 10.43┬▒8.97 | 23.01┬▒18.78 | 40.86┬▒23.00 | 40.74┬▒23.09 | 0.029* | 0.002* | 0.001* | ||||
| Muscle thickness | |||||||||||
| ŌĆāTrA (mm) | Ho | 2.93┬▒0.67 | 2.94┬▒0.61 | 3.11┬▒0.57 | 3.10┬▒0.84 | 0.153 | 0.561 | 0.568 | 1.000 | 0.199 | 0.530 |
| Ve | 2.86┬▒0.44 | 2.88┬▒0.36 | 2.97┬▒0.40 | 2.86┬▒0.40 | 1.000 | 1.000 | 1.000 | ||||
| ŌĆāMulti (mm) | Ho | 23.29┬▒4.25 | 23.67┬▒3.91 | 23.73┬▒3.33 | 23.37┬▒2.52 | 0.737 | 0.380 | 0.906 | 1.000 | 1.000 | 1.000 |
| Ve | 22.83┬▒2.57 | 23.05┬▒2.17 | 23,48┬▒2.13 | 22.84┬▒2.57 | 1.000 | 1.000 | 1.000 | ||||
| Balance control | |||||||||||
| ŌĆāAP | Ho | 3.86┬▒1.23 | 2.96┬▒0.65 | 2.38┬▒0.71 | 2.50┬▒0.67 | <0.001 | <0.001 | 0.647 | 0.017* | 0.000* | 0.001* |
| Ve | 4.00┬▒0.80 | 3.11┬▒0.89 | 2.52┬▒0.71 | 2.41┬▒0.65 | 0.000* | 0.000* | 0.000* | ||||
| ŌĆāML | Ho | 3.83┬▒1.26 | 2.58┬▒0.97 | 2.23┬▒0.79 | 2.28┬▒0.71 | <0.001 | <0.001 | 0.522 | 0.104 | 0.005* | 0.014* |
| Ve | 3.89┬▒1.79 | 3.02┬▒1,27 | 2.25┬▒0.79 | 2.19┬▒0.71 | 0.008* | 0.001* | 0.010* | ||||
Values are presented as mean┬▒standard deviation.
HVE, horizontal vibration exercise; VVE, vertical vibration exercise; Ho, horizontal vibration group; Ve, vertical vibration group; t0, assessment at baseline; t1, assessment at 6 weeks; t2, assessment at 12 weeks; t3, assessment performed after 4 weeks from the end of intervention; VAS, visual analogue scale; ODI, Oswestry disability index; LFPT, lumbar flexor peak torque; LEPT, lumbar extensor peak torque; LFAP, lumbar flexor average power; LEAP, lumbar extensor average power; TrA, transverse abdominis; Multi, multifidus; AP, anterior-posterior; ML, medial-lateral.
REFERENCES
1. Krismer M, van Tulder M; Low Back Pain Group of the Bone and Joint Health Strategies for Europe Project. Strategies for prevention and management of musculoskeletal conditions. Low back pain (non-specific). Best Pract Res Clin Rheumatol 2007;21:77-91.


2. Trapp W, Weinberger M, Erk S, Fuchs B, Mueller M, Gallhofer B, et al. A brief intervention utilising visual feedback reduces pain and enhances tactile acuity in CLBP patients. J Back Musculoskelet Rehabil 2015;28:651-60.


3. Bagheri R, Takamjani IE, Dadgoo M, Sarrafzadeh J, Ahmadi A, Pourahmadi MR, et al. A protocol for clinical trial study of the effect of core stabilization exercises on spine kinematics during gait with and without load in patients with non-specific chronic low back pain. Chiropr Man Therap 2017;25:31.




4. Graves JE, Pollock ML, Foster D, Leggett SH, Carpenter DM, Vuoso R, et al. Effect of training frequency and specificity on isometric lumbar extension strength. Spine (Phila Pa 1976) 1990;15:504-9.


5. Gill KP, Callaghan MJ. The measurement of lumbar proprioception in individuals with and without low back pain. Spine (Phila Pa 1976) 1998;23:371-7.


6. Iverson BD, Gossman MR, Shaddeau SA, Turner ME Jr. Balance performance, force production, and activity levels in noninstitutionalized men 60 to 90 years of age. Phys Ther 1990;70:348-55.



7. Wang XQ, Pi YL, Chen PJ, Chen BL, Liang LC, Li X, et al. Whole body vibration exercise for chronic low back pain: study protocol for a single-blind randomized controlled trial. Trials 2014;15:104.



8. Chen H, Ma J, Lu B, Ma XL. The effect of whole-body vibration training on lean mass: a PRISMA-compliant meta-analysis. Medicine (Baltimore) 2017;96:e8390.



9. Park SY, Son WM, Kwon OS. Effects of whole body vibration training on body composition, skeletal muscle strength, and cardiovascular health. J Exerc Rehabil 2015;11:289-95.




10. Bagheri J, van den Berg-Emons RJ, Pel JJ, Horemans HL, Stam HJ. Acute effects of whole-body vibration on jump force and jump rate of force development: a comparative study of different devices. J Strength Cond Res 2012;26:691-6.


11. del Pozo-Cruz B, Hernandez Mocholi MA, Adsuar JC, Parraca JA, Muro I, Gusi N. Effects of whole body vibration therapy on main outcome measures for chronic non-specific low back pain: a single-blind randomized controlled trial. J Rehabil Med 2011;43:689-94.


12. Rittweger J, Just K, Kautzsch K, Reeg P, Felsenberg D. Treatment of chronic lower back pain with lumbar extension and whole-body vibration exercise: a randomized controlled trial. Spine (Phila Pa 1976) 2002;27:1829-34.


13. Sitja Rabert M, Rigau Comas D, Fort Vanmeerhaeghe A, Santoyo Medina C, Roque i Figuls M, Romero-Rodriguez D, et al. Whole-body vibration training for patients with neurodegenerative disease. Cochrane Database Syst Rev 2012;(2): CD009097.


14. Kaeding TS, Karch A, Schwarz R, Flor T, Wittke TC, Kuck M, et al. Whole-body vibration training as a workplace-based sports activity for employees with chronic low-back pain. Scand J Med Sci Sports 2017;27:2027-39.


15. Gillan SN, Sutherland S, Cormack TG. Vitreous hemorrhage after whole-body vibration training. Retin Cases Brief Rep 2011;5:130-1.


16. Vela JI, Andreu D, Diaz-Cascajosa J, Buil JA. Intraocular lens dislocation after whole-body vibration. J Cataract Refract Surg 2010;36:1790-1.


17. Kiiski J, Heinonen A, Jarvinen TL, Kannus P, Sievanen H. Transmission of vertical whole body vibration to the human body. J Bone Miner Res 2008;23:1318-25.


18. Lee G. Does whole-body vibration training in the horizontal direction have effects on motor function and balance of chronic stroke survivors? A preliminary study. J Phys Ther Sci 2015;27:1133-6.



19. Shim C, Lee Y, Lee D, Jeong B, Kim J, Choi Y, et al. Effect of whole body vibration exercise in the horizontal direction on balance and fear of falling in elderly people: a pilot study. J Phys Ther Sci 2014;26:1083-6.



20. Jung IG, Yu IY, Kim SY, Lee DK, Oh JS. Reliability of ankle dorsiflexion passive range of motion measurements obtained using a hand-held goniometer and Biodex dynamometer in stroke patients. J Phys Ther Sci 2015;27:1899-901.



21. Sweeney N, OŌĆÖSullivan C, Kelly G. Multifidus muscle size and percentage thickness changes among patients with unilateral chronic low back pain (CLBP) and healthy controls in prone and standing. Man Ther 2014;19:433-9.


22. Nuzzo JL, Haun DW, Mayer JM. Ultrasound measurements of lumbar multifidus and abdominal muscle size in firefighters. J Back Musculoskelet Rehabil 2014;27:427-33.


23. Sohn MK, Lee SS, Song HT. Effects of acute low back pain on postural control. Ann Rehabil Med 2013;37:17-25.



24. Glave AP, Didier JJ, Weatherwax J, Browning SJ, Fiaud V. Testing postural stability: are the star excursion balance test and Biodex Balance System limits of stability tests consistent? Gait Posture 2016;43:225-7.


25. Kakigi R, Shibasaki H. Mechanisms of pain relief by vibration and movement. J Neurol Neurosurg Psychiatry 1992;55:282-6.



26. Shilpapriya M, Jayanthi M, Reddy VN, Sakthivel R, Selvaraju G, Vijayakumar P. Effectiveness of new vibration delivery system on pain associated with injection of local anesthesia in children. J Indian Soc Pedod Prev Dent 2015;33:173-6.


27. Torvinen S, Kannus P, Sievanen H, Jarvinen TA, Pasanen M, Kontulainen S, et al. Effect of four-month vertical whole body vibration on performance and balance. Med Sci Sports Exerc 2002;34:1523-8.


28. Perchthaler D, Grau S, Hein T. Evaluation of a six-week whole-body vibration intervention on neuromuscular performance in older adults. J Strength Cond Res 2015;29:86-95.


29. Baard ML, Pietersen J, van Rensburg SJ. Interventions for chronic low back pain: whole body vibration and spinal stabilisation. S Afr J Sports Med 2011;23:35-9.

30. Torabi M, Okhovatian F, Naimi SS, Baghban AA, Vafaee R. The effect of core stability training with and without whole body vibration in chronic low back pain patients. J Paramed Sci 2013;4:17-25.
31. Hagbarth KE, Hongell A, Wallin BG. The effect of gamma fibre block on afferent muscle nerve activity during voluntary contractions. Acta Physiol Scand 1970;79:27. A-28A.
32. Yang J, Seo D. The effects of whole body vibration on static balance, spinal curvature, pain, and disability of patients with low back pain. J Phys Ther Sci 2015;27:805-8.



33. Tsaklis PV, Grooten WJ, Franzen E. Effects of weightshift training on balance control and weight distribution in chronic stroke: a pilot study. Top Stroke Rehabil 2012;19:23-31.


34. Knuttgen HG. Strength training and aerobic exercise: comparison and contrast. J Strength Cond Res 2007;21:973-8.


35. Kurz I, Gimmon Y, Shapiro A, Debi R, Snir Y, Melzer I. Unexpected perturbations training improves balance control and voluntary stepping times in older adults: a double blind randomized control trial. BMC Geriatr 2016;16:58.



- TOOLS
-
METRICS

- Related articles in ARM
-
The Clinical Effect of Facet Joint Injection in Chronic Low Back Pain Patients1996 March;20(1)






