The Effects of Neuromuscular Electrical Stimulation on Cardiopulmonary Function in Healthy Adults
Article information
Abstract
Objective
To evaluate the effect of neuromuscular electrical stimulation (NMES) on cardiopulmonary function in healthy adults.
Method
Thirty-six healthy adults without a cardiac problem were enrolled. All patients were randomly assigned to either a control (17 subjects, mean age 29.41) or an electrical stimulation group (19 subjects, mean age 29.26). The electrical stimulation group received NMES on both sides of quadriceps muscle using a Walking Man II® in a sitting position for 30 minutes over 2 weeks. Maximum oxygen consumption (VO2max), metabolic equivalent (MET), resting, maximal heart rate (RHR, MHR), resting, maximal blood pressure (RBP, MBP), and maximal rate pressure product (MRPP), exercise tolerance test (ETT) duration were determined using an exercise tolerance test and a 6 minute walk test (6MWT) before and after treatment.
Results
The electrical stimulation group showed a significant increase in VO2max (p=0.03), 6MWT (p<0.01), MHR (p<0.04), MsBP (p<0.03), ETT duration (p<0.01) and a significant decrease in RsBP (p<0.02) as compared with the control group after two weeks. NMES induced changes improved only in RsBP (p<0.049) and ETT duration (p<0.01). The effects of NMES training were stronger in females.
Conclusion
We suggest that NMES is an additional therapeutic option for cardiopulmonary exercise in disabled patients with severe refractory heart failure or acute AMI.
INTRODUCTION
Cardiovascular diseases are the third leading cause of death after cancer and cerebrovascular disease according to the annual data issued by the Korean National Statistical Office in 2010, with a mortality rate of 46.9 per 100,000 members of the population per year.1 In those under fifty heart disease is the leading cause of death.
The importance and beneficial effect of cardiac rehabilitation is well documented for a variety of cardiac disorders. Cardiac rehabilitation improves cardiopulmonary function and returns the patient to an optimal level of physical, psychological, social well-being. Furthermore, it also reduces heart attack recurrence and lowers mortality rates.2-4
Despite the numerous benefits of cardiac rehabilitation programs, participation is still very low in Korea, because of lack of sufficient cardiac monitoring equipment as well as patients' barriers to care including occupational issues, a lack of interest in rehabilitation, a reluctance to make lifestyle changes, depression, or a lack of family cooperation.5 In cardiac rehabilitation, it is very important to continue aerobic exercise for several weeks post-illness, but many patients have problems in performing exercise because of both cardiac origin causes and non-cardiac reasons such as fatigue, dyspnea, psychiatric and neuromuscular problems.6 Lower limb muscular dysfunction and fatigue in particular contribute to poor treadmill performance.7-9
Neuromuscular electrical stimulation (NMES) exercise can increase strength and prevent atrophy of paralyzed muscles using stimulation patterns that induce repetitive contractions in selected muscle groups. Clinical application of NMES offers functional and therapeutic benefits. Functional tasks include standing or ambulatory activities, upper-limb performance during activities required for daily life, and the control of respiration and bladder function in stroke or SCI patients.10,11 NMES is also used for therapeutic purposes including motor relearning, cardiovascular conditioning, treatment of spasticity, and prevention of muscle atrophy, disuse osteoporosis, and deep venous thrombosis (DVT).12
NMES could be an alternative strategy to increase the muscle work performed in severely disabled patients. Numerous investigations have shown its muscular benefits in patients with muscle weakness associated with atrophy and intact innervation. More recent studies have shown an interesting beneficial effect of NMES in patients with stroke and chronic heart failure in terms of strength and endurance improvement, as well as central circulatory adaptations and enhanced aerobic capacity. However, there have been few studies in Korea, and limited data in healthy persons, or severely deconditioned patients with AMI. The present study was undertaken to evaluate the effect of neuromuscular electrical stimulation (NMES) on cardiopulmonary function in healthy adults without any cardiac problems using an exercise stress test, and to apply the basic data for severely deconditioned patients with AMI in Korea.
MATERIALS AND METHODS
Subjects
The research participants were 36 healthy adults without a cardiac problem who volunteered for the study. All study subjects provided informed consent before the selection procedure. Out study protocol was reviewed and approved by the University of Jeju Institutional Review Board, and that written informed consent for this research project was obtained from each participant. The 36 participants had no history of cardiopulmonary disease, stroke, or of a neurologic condition. Radiological and clinical examinations of the lower limbs were performed to confirm the absence of fractures, joint degeneration changes, or clinical joint instability. In addition, no participant had a pacemaker fitted, skin damage, or skin ulcers.
Methods
All participants were randomly assigned to either the control or the electrical stimulation groups. Members of the electrical stimulation group (19 subjects, 10 male, mean age 29.26±2.92) received NMES on both sides of quadriceps muscle using a Walking Man II® (Cyber Medic, Seoul, Korea) in sitting position for 30 minutes for 2 weeks. Stimulation protocol consisted of a symmetrical biphasic square pulse at 35 Hz, a duty cycle of 10 s on and 12 s off, a pulse time of 250 µsec during each 30 min treatment session.13 The maximum power output of the stimulator was 100 mA. The stimulation intensity was increased within the range of motor threshold to maximal comfortably tolerated intensity. The control group (17 subjects, 6 male, mean age 29.41±3.80) received electrical stimulation at the sensory threshold without muscle contraction for same period of NMES group.
Both groups underwent a symptom limited exercise test using a Bruce protocol. The exercise test was performed using a CH2000 Integrated 12-lead Stress Testing ECG (Cambridge Heart, Tewksbury, USA), Quark CPET Breath Gas Exchange data analysis (Cosmed, Rome, Italy), a T-2100 treadmill (GE, Fairfield, USA), and a BP (blood pressure) and HR (heart rate) monitor (Fig. 1).
(A) Neuromuscular electrical stimulation Walking Man II® (Cyber Medic, Seoul, Korea), (B) Electrode placement on quadriceps muscles on both sides, (C) Cardiopulmonary function test.
Maximum oxygen consumption (VO2max), metabolic equivalent (MET), resting, maximal heart rate (RHR, MHR), resting, maximal blood pressure (RBP, MBP), and rate pressure product (MRPP), exercise tolerance test (ETT) duration were determined using an exercise tolerance test. In addition, functional exercise capacity was measured by the 6-minute walk test (6MWT) in a 50 m in-hospital corridor, performed before and after testing.
Statistical analysis
Statistical analysis was performed using SPSS for Windows version 14.0 (SPSS V14.0K, SPSS Inc., Chicago, IL, USA). The Chi-square and Wilcoxon's rank sum tests were used to compare group baseline characteristics, and Wilcoxon's rank sum test was also used to compare group cardiopulmonary functions at baseline and 2 weeks after testing. The differences between group indicators at baseline and 2 weeks after testing were compared using the Mann-Whitney test. Null hypotheses of no difference were rejected if p-values were less than 0.05.
RESULTS
General characteristics of the participants
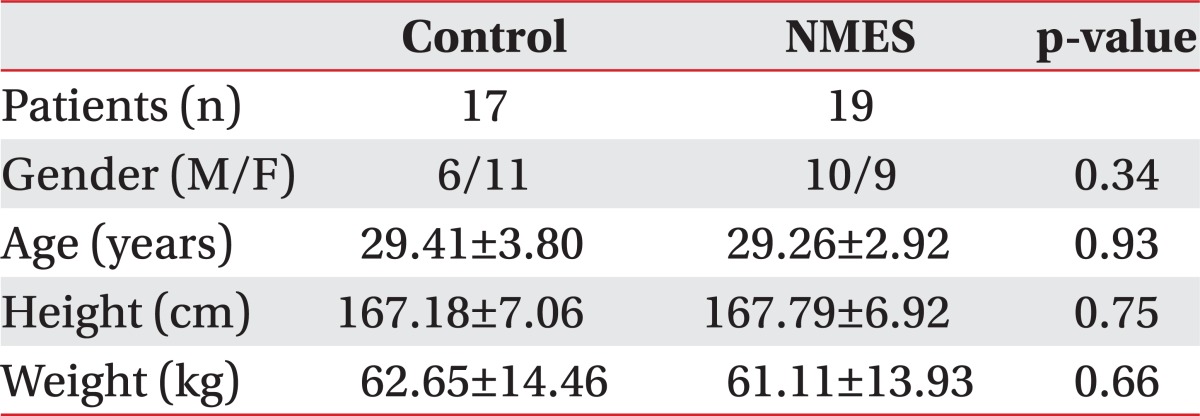
The baseline characteristics of the participants are summarized in Table 1. Participants were assigned to the experimental (19 subjects, 10 male, mean age 29.26±2.92) or the control group (17 subjects, 6 male, mean age 29.41±3.80).
Demographic characteristics including gender, age, height, weight and baseline cardiopulmonary functions of these two groups were not significantly different (Table 1).
Changes in cardiopulmonary function parameters at 2 weeks after testing in each group
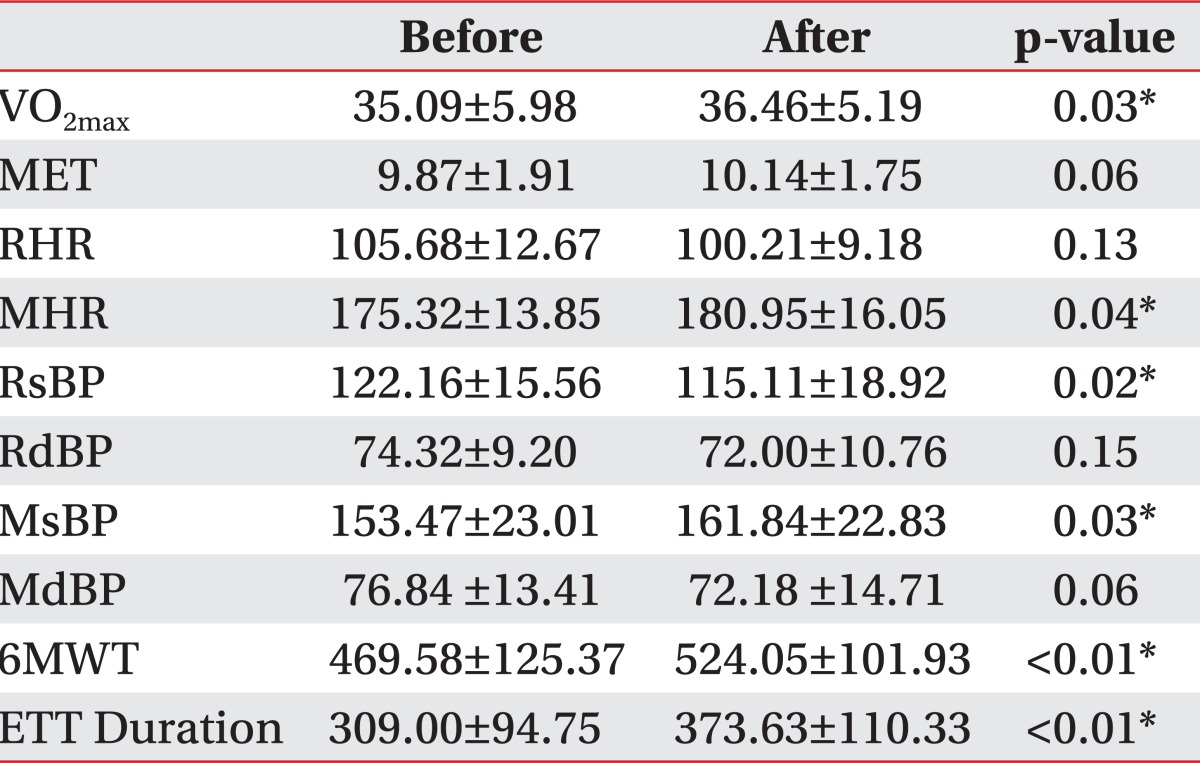
In the control group, cardiopulmonary function parameters before and at 2 weeks after testing were similar. However, in the NMES group, maximum oxygen consumptions (VO2max) (p=0.03) significantly increased from 35.09±5.98 to 36.46±5.19, and 6-minute walk test (6MWT) (p<0.01) increased from 469.58±125.37 m to 524.05±101.93 m. Resting systolic blood pressures (RsBP) (p=0.02) decreased from 122.16±15.56 to 115.11±18.92. Maximal heart rates (MHR) (p=0.04), maximal systolic blood pressure (MsBP) (p=0.03), exercise tolerance test (ETT) duration (p<0.01) also increased significantly, but metabolic equivalents (MET), resting heart rates (RHR), resting diastolic blood pressure (RdBP), maximal diastolic blood pressures (MdBP) showed no significant change (Table 2).
Comparison of the changes in cardiopulmonary function parameters in the two groups at 2 weeks after testing
When effects before and 2 weeks after testing were compared in the each groups, resting systolic blood pressure (RsBP) decreased significantly in the NMES group (-7.05±11.95, p=0.049), and exercise tolerance test (ETT) duration increased (64.63±72.27, p=0.01). However, no significant differences were found for changes of maximum oxygen consumptions (VO2max), 6-minute walk test (6MWT), metabolic equivalents (MET), resting, maximal heart rates (RHR, MHR), resting diastolic blood pressure (RdBP), maximal blood pressures (MsBP, MdBP). The 6-minute walk test distance (6MWT) tended to increase in the NMES group (p=0.10).
Comparison of the changes in cardiopulmonary function parameters according to gender between the two groups at 2 weeks after testing
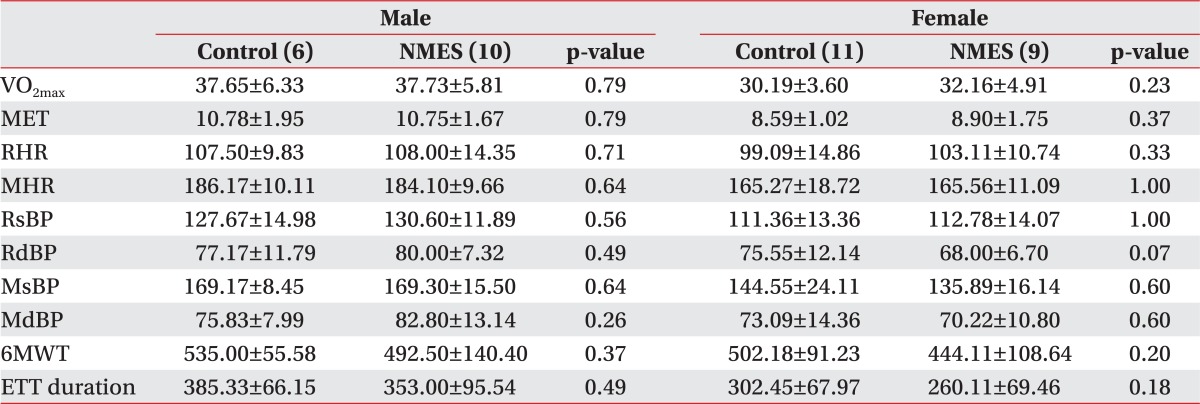
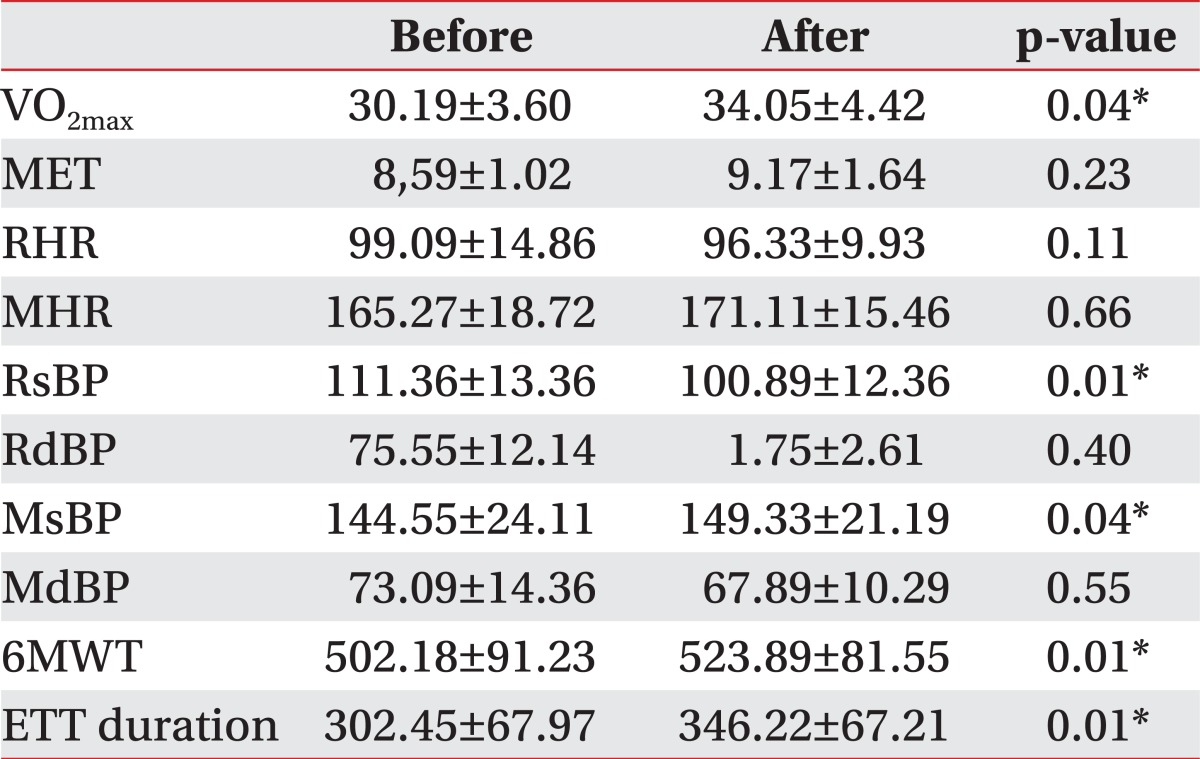
Both groups were analyzed by gender, in order to compare the NMES effects by gender. Neither male group nor female group differed in cardiopulmonary function between the two groups at baseline (Table 3). In the female NMES group, maximum oxygen consumptions (VO2max) (p=0.04), maximal systolic blood pressure (MsBP) (p=0.04), exercise tolerance test (ETT) duration (p=0.01), 6-minute walk test (6MWT) (p=0.01) increased significantly after two weeks. Resting systolic blood pressure (RsBP) (p=0.01) decreased from 111.36±13.36 to 100.89±12.36 (Table 4). Changes in the female NMES group were significantly improved, compared with the female control group in RsBP (p=0.03), ETT duration (p=<0.01), 6-minute walk test (p=0.01). However, the male NMES group did not differ in cardiopulmonary function, compared with male control group after 2 weeks (Table 5).
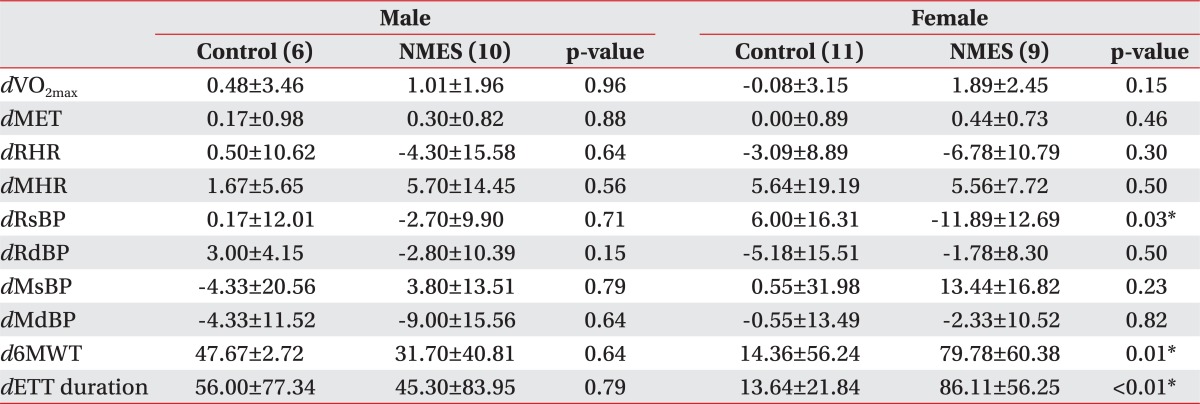
Comparison of Change in Cardiopulmonary Function Parameters by Gender between Groups at 2 Weeks after Testing
During cardiopulmonary function testing in the study, no cardiopulmonary complication, such as angina pectoris, AMI, or death, occurred in either group.
DISCUSSION
The results of this study show that NMES training in healthy adults increased cardiopulmonary functional capacity measured by maximum oxygen consumptions (VO2max), 6-minute walk test (6MWT), resting systolic blood pressures (RsBP), maximal heart rates (MHR), maximal systolic blood pressure (MsBP). Several studies have reached similar conclusions in patients with CHF, COPD, SCI, and stroke patients.9 Vaquero et al.14 reported significant increases in peak workload and peak oxygen uptake (VO2max) in the NMES group in cardiac transplantation patients. In addition, Quittan et al.15 reported significant improvements in muscle strength and reduction of fatigue in chronic heart failure. Karavidas et al.16 also reported significant improvements in the 6-minute walk test (6MWT). These studies shows positive effects on exercise capacity, muscle strength in the NMES group, compared with control groups who underwent usual care or sham stimulation. Our study also showed improvement in maximum oxygen consumptions (VO2max) and 6-minute walk test (6MWT) within NMES group after 2 weeks, although NMES-induced changes improved only in RsBP and ETT duration.
In addition, according to Stuart Harris et al.17 and Dobsak et al.,18 NMES training produced beneficial change were not prominent but similar to aerobic exercise such as bicycle training in exercise capacities including the 6-min walk test, exercise time, muscle performance. So our conclusion suggests that NMES is an alternative to classical aerobic exercise training in CHF patients. Besides applying NMES in CHF patients, Neder et al.19 reported, NMES training resulted in significant improvements in muscle strength and exercise tolerance in COPD patients. Vivodtzev et al.20 also reported a significant improvement in maximal voluntary contraction of the quadriceps muscle and a decrease in the dyspnea when NMES was combined with rehabilitation for COPD patients.
Contractions of leg muscles stimulates leg muscle venous pump activity, increasing venous return, stroke volume, and cardiac output via the Frank-starling mechanism.21 NMES increases strength and muscle endurance by inducing hypertrophy and histochemical changes in muscles, by increasing capillary density, mitochondrial concentrations, and oxidative enzyme levels, associated with the transformation of muscle fiber.12,18,22,23 These changes increase aero-metabolic capacity, prevent of muscle atrophy, and increase resistance to fatigue. That is, it improves physical capacity and the abilities of patients to perform daily activities and reduce medical complications.21 Due to the short duration of our study period it was difficult to produce enough changes in muscle strength or histological morphology. Also the absence of isokinetic equipment for checking muscle strength disabled us from evaluating the change of muscle strength. Nevertheless, the increase in ETT duration demonstrates the increment in fatigue resistance and improved cardiopulmonary function.
As oxygen uptake level depends on stroke volume (which increases O2 delivery to exercising muscles) and the abilities of skeletal muscles to consume O2, increases in VO2 values reflect functional capacity improvements. Banerjee et al.24 reported that electrical stimulation was capable of producing a physiological response consistent with cardiovascular exercise at mild to moderate intensities. He suggested that the NMES-induced level of energy expenditure was similar to the level expected for activities such as walking 3-3.5 mph.
Hsu et al.25 reported that NMES only induced approximately such as 76 kcal/h on total calories and 0.26 L/min on oxygen consumption. It achieves this without producing movement or voluntary exercise such as ergometer or treadmill training. These findings supported the hypothesis that NMES is a useful tool for improving cardiopulmonary function and physical activity for those unable to participate in exercise and are in line with the results of our study.
There are several limitations in this study. The most serious is the small number of subjects evaluated, insufficient to monitor the gender effects on NMES training, although the female NMES group revealed the only significant improvement after two weeks. Second, although our study revealed improvement in maximum oxygen consumptions (VO2max) and 6-minute walk test within NMES group after 2 weeks, there was no significant change of variance between two groups except resting systolic blood pressure (RsBP) and ETT duration. The therapeutic intervention period of 2-weeks might be too short to establish the sufficient effects of NMES training, compared with control. And third, we could not evaluate the change of quadriceps muscle strength, because they were normal grade in MRC at baseline. The absence of isokinetic equipment made it impossible to check the subtle change of muscle strength. Fourthly, our participants did not perform aerobic exercise. Therefore, the improvement of exercise capacities was milder than that of previous studies. Finally, we decided the intensity as motor threshold, under the consideration of 2-week treatment and volunteers' endurance and compliance. However it could be regarded that the higher intensity of NMES caused a greater improvement in the basis of several studies. Accordingly, further large scale, long term follow up studies considering NMES intensity and gender should be conducted to examine cardiopulmonary function capacity improvements after NMES exercise.
CONCLUSION
NMES proved to be effective, safe therapy to enhance exercise capacity and functional ability. This study shows NMES training is capable of increasing the cardiopulmonary responses in healthy adults. So we suggest that NMES training is viewed as a potential therapeutic option for cardiopulmonary rehabilitation. Although our study did not include aerobic exercise, cardiopulmonary function is improved. NMES was more effective in females with poor baseline functionality. So we propose that NMES training is a useful adjunct to medical therapy in disabled patients with severe refractory heart failure or acute AMI.
ACKNOWLEDGEMENTS
This work was supported by a research grant from Jeju National University Hospital.