INTRODUCTION
Traumatic brain injury (TBI) is one of the common causes of disability in adults, and is associated with longterm or lifelong physical, cognitive, behavioral, and emotional consequences [
1]. Swallowing impairment is a significant issue in the management of patients with TBI because many different mechanisms, such as impaired oropharyngeal function, cognitive deficit, and behavioral problems, can affect their swallowing [
2,
3,
4]. Among inpatient TBI patients in rehabilitation, the incidence of impaired oral feeding reportedly ranges between 25% to 93% [
2,
3,
5,
6]. In particular, 37% of severe TBI patients are unable to recover to an unrestricted diet after 18 weeks of inpatient rehabilitation [
7]. Since dysphagia after brain injury can lead to malnutrition, dehydration, and aspiration pneumonia, adequate management of dysphagia is an essential part of TBI rehabilitation.
A few studies have investigated the characteristics of dysphagia in TBI patients [
2,
4,
5,
8]. Prolonged oral transit, delayed swallowing reflex, and altered lingual control are frequently reported in patients with TBI [
5,
8]. However, a prospective study has demonstrated that pharyngeal dysfunctions, including aspiration, are very frequent in severe TBI patients [
4]. Additionally, the Rancho Los Amigo Scale, computerized tomography (CT) scans, ventilation time, and aspiration are reported as significant independent predictors of impaired oral intake in TBI patients [
3,
4,
9]. Although these findings suggest a direction of dysphagia management in TBI patients, relevant information is still lacking.
In contrast, numerous studies have investigated the characteristics and management of dysphagia in patients with stroke. A Cochrane review reported that behavioral interventions and acupuncture reduced dysphagia, and pharyngeal electrical stimulation reduced pharyngeal transit time in stroke patients [
10]. Conventional treatments such as change in posture and position for swallowing, learning new swallowing maneuvers, oral stimulation, neuromuscular electrical stimulation, and alteration in food amounts and texture have been recommended as treatment options [
11]. These have been adopted for the treatment of dysphagic TBI patients without specific evidence of their effectiveness. These therapeutic approaches will require modification for TBI patients if dysphagia characteristics differ between TBI and stroke patients.
Therefore, the purpose of this study was to compare swallowing characteristics between TBI and stroke patients with dysphagia through the assessment of videofluoroscopic swallowing study (VFSS) findings. We hypothesized that frequent abnormal findings in oropharyngeal swallowing differ between the two patient groups.
RESULTS
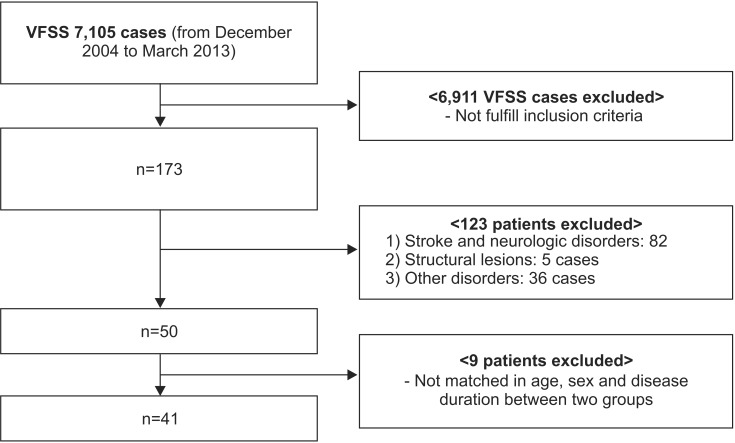
Demographic and clinical patient characteristics
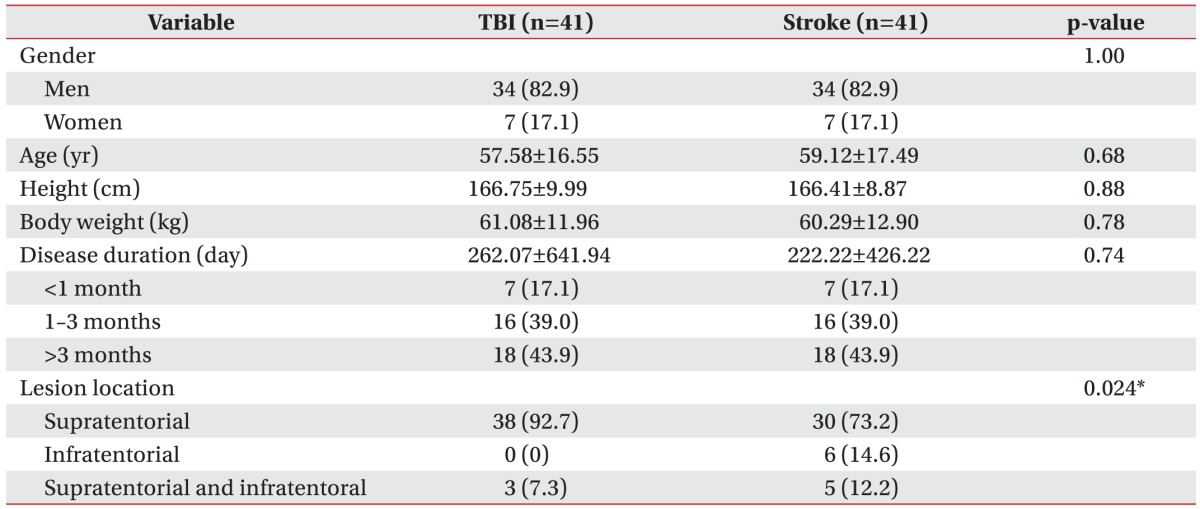
Mean (Вұstandard deviation) patient age was 57.6Вұ16.6 years (range, 24вҖ“88) and 59.1Вұ17.5 years (range, 23вҖ“86) in the TBI and stroke groups, respectively. All demographic data were presented in
Table 1. There was no significant difference in age, sex, height, and body weight between the two groups. Disease duration in the TBI and stroke group was 262.1Вұ641.9 days and 222.2Вұ426.2 days, respectively. The number of the patients classified at the acute, sub-acute, and chronic stage was 7 (17.1%), 16 (39.0%), and 18 (43.9%), respectively. The location of lesions differed significantly between the two groups (p=0.024) (
Table 1).
Dysphagia severity and characteristics in TBI and stroke patients
The mean severity of dysphagia between the two patient groups showed no significant difference according to the ASHA NOMS swallowing scale (3.6Вұ2.2 in TBI vs. 3.8Вұ2.2 in stroke; p=0.600) and the VDS (35.3Вұ21.0 in TBI vs. 34.2Вұ20.4 in stroke; p=0.824) (
Table 2).
The VFSS findings showed no significant difference between the two groups. In patients with TBI, the most common VFSS findings, in descending order, included aspiration or penetration (73.2%), decreased laryngeal elevation (68.3%), and reduced epiglottis inversion (61%). In stroke patient group, aspiration or penetration (73.2%), reduced epiglottis inversion (68.3%), and decreased laryngeal elevation (63.4%) were the most common VFSS findings. Furthermore, the mean CDS showed no significant difference between the two groups (17.2Вұ16.7 in TBI vs. 19.8Вұ19.1 in stroke; p=0.538).
Impact of surgical intervention
Among the 41 TBI patients, 20 underwent surgical intervention. Of these patients, 12 underwent craniectomy and 6 underwent craniotomy. The remaining 2 patients had undergone brain surgery; however, due to the absence of medical records, we could not determine the type of surgery. Of the 20 patients with surgical intervention, 8 patients could eat orally (ASHA NOMS swallowing scale вүҘ4), and of the 21 patients without surgical intervention, 17 patients could eat orally. Dysphagia severity showed significant difference between patients with surgical intervention and those without when assessed using the ASHA NOMS swallowing scale вүҘ4 (oral-feeder) (p=0.011). However, CDS, VDS, OTT, PDT, and PTT showed no significant difference between these patient groups.
Impact of radiological lesion
Common radiological findings in the TBI patient group comprised subdural hemorrhage (SDH; 60.98%), intracranial hemorrhage (ICH; 43.90%), subarachnoid hemorrhage (SAH; 34.15%), extradural hematoma (EDH; 21.95%), and intraventricular hemorrhage (IVH; 12.20%). Thirteen patients (31.71%) exhibited bilateral brain lesion, 14 (34.15%) patients' radiological findings presented as a midline shift, and 3 (7.32%) patients had a brainstem lesion. The outcome measures showed no significant differences between patients with each radiological lesion, including SDH, ICH, SAH, EDH, IVH, bilateral brain lesion, midline shift, and brainstem involvement and those without a lesion.
DISCUSSION
To our knowledge, this is the first study to compare swallowing characteristics and severity of dysphagia between patients with TBI and stroke. There was no significant difference in the severity and characteristics of dysphagia between the two patient groups. In the TBI group, patients who underwent surgical intervention showed a significant positive relationship with non-oral feeding.
Although no previous studies have compared dysphagia between TBI and stroke patients, comparative studies have been conducted on mobility differences between patients with TBI and those with stroke. Juneja et al. [
15] reported no differences in the effects of admission and discharge status on balance and activities of daily living function between patients with TBI and those with stroke. In contrast, Eng et al. [
16] found that patients with TBI were admitted and discharged with a higher mobility status, as compared with patients with stroke; however, the actual change in mobility status (improvement) did not differ between the two patient groups. The significant difference in age partially accounted for the differences between the TBI and stroke patient groups. However, the admission and discharge mobility status differed significantly between these two patient groups in the previous studies. These differences are possibly due to the specific pathology of the different brain injuries and their resulting clinical manifestations. In our study, although there was significant difference in lesion location, all the TBI patients had intracranial lesion as in stroke patients. Major portion of lesion location in both groups was supratentorial lesion (TBI, 92.7%; Stroke, 73.1%). Thus, this pathologic similarity might attribute to the similar characteristics of dysphagia between the two groups. In addition, the common VFSS findings in our study, such as aspiration or penetration, decreased laryngeal elevation and reduced epiglottis inversion, could be shown in both infratentorial and supratentorial lesion.
In our study, common VFSS findings in TBI patients comprised aspiration or penetration, decreased laryngeal elevation, and reduced epiglottis inversion. This suggests that dysphagia in the TBI patients mainly resulted from pharyngeal dysfunction. This result supports the finding of a previous study, which demonstrated frequent pharyngeal dysfunctions, including aspiration, in severe TBI patients [
4]. In contrast, other studies have reported oral phase abnormality as the most common VFSS finding in TBI patients [
4,
8,
17]. In these studies, patients were young adults (average age ranged between 26 and 34 years); a younger mean age than the TBI patients in the present study (mean age, 57.58 years). Mean disease duration in the previous studies was between 17.6 days and 2.5 months, while in the present study it was approximately 262 days. Although the effect of age is unclear, short disease duration could affect oral phase abnormality. Due to cognitive dysfunction or consciousness disorder, patients in early TBI stage might be at higher risk of oral dysfunction than patients in our study.
Mackay et al. [
3] reported an ICH incidence of 25.9%, which is lower than the ICH incidence in the present study (43.9%). The presence of ICH is associated with traumatic parenchymal lesion. Larger lesions may place the patients at risk of further neurological deterioration, herniation, and death [
17]. In addition, the incidence of patients with brain stem hemorrhage in the present study was higher (7.31%) than previously reported (mean, 1.63%) [
18]. Brain stem lesions can result in abnormal swallowing pattern generation or abnormal distribution of the swallowing drive to the various motor neuronal pools [
19,
20]. This suggests that patients in the present study were at higher risk of pharyngeal dysfunction than patients in the previous study.
Terre and Mearin [
8] showed that while frequent early VFSS findings revealed an increase in OTT and alterations in lingual control, they were normalized in 80% of patients at 1 year of follow-up. In relation to temporary dysphagia, most cases can be categorized as contusion or brain cortex lesion, and present with oral phase abnormality due to cognitive problems or attention deficiency [
3,
4,
6,
17]. In these cases, swallowing function can recover with the recovery of cognitive function. In contrast, pharyngeal dysfunction has the potential to be main mechanism in cases of persistent dysphagia.
The subgroup analyses suggested that patients with surgical intervention might have a greater chance of nonoral feeding (ASHA NOMS scale <4) than those without. However, none of the radiological findings were statistically related with dysphagia severity and characteristics. A previous study reported that severe CT findings (lesions that require surgical intervention, midline shift, or brain stem involvement) were related to abnormal swallowing in TBI patients [
4]. This supports the present study's findings that surgical intervention can affect swallowing function. The indications of surgical intervention were low Glasgow Coma Scale (GCS) or increased intracranial pressure [
21]. Low GCS could be caused by brain stem lesion involving reticular formation or bilateral frontal lesion, and increased intracranial pressure could be caused by extensive hemorrhage. These pathologies may negatively impact swallowing function.
In 2013, Stevens and Sutter [
22] reported that SAH and IVH were prognostic factors of TBI. However, the present study's results suggest that there is no direct relationship between radiologic lesion and dysphagia severity or characteristics. While brain stem lesions have been reported to cause severe dysphagia in stroke patients, there have been few studies on dysphagia associated with brain stem lesion in TBI patients [
23]. As discussed previously, the incidence of brain stem lesions was lower than 10% in the present study. Therefore, the influence of brain stem involvement on dysphagia in TBI patients may have been underestimated due to the low incidence of those lesions. Therefore, patients who undergo surgical intervention after TBI require more careful care in relation to dysphagia.
This study had some limitations. Firstly, the target population of this study comprised severe TBI patients with dysphagia. Of the 41 patients, 20 patients underwent brain surgery; however, a previous study reported that out of a total of 816 TBI patients, only 40 patients underwent decompressive craniectomy [
21]. This suggests that a higher proportion of patients with more severe TBI or those with high intracranial pressure at first onset may have been included in the present study, as compared to previous research. Additionally, almost all patients originated from another first to tertiary care center, and the study population was not from a general population; only patients who underwent VFSS were included. Furthermore, all patients exhibited a radiologic lesion on the brain. Therefore, the findings of our study are limited to the generalized TBI patient population. Secondly, the duration of disease was heterogeneous. The shortest disease duration was 9 days after onset and the longest was 1,494 days after TBI onset, with a standard deviation of 641.9 days. Due to the variability of disease duration, it is difficult to confirm if the outcomes of this study consistently reflect the physiology of TBI. Finally, the clinical information, including the severity of TBI and intracranial pressure, was insufficient. Severity of TBI measured using the GCS or the Rancho Los Amigo Scale has been reported to be related to the incidence of dysphagia [
3,
4,
6,
17]. However in this study, such information was missing because patients with chronic TBI were included. Despite these limitations, information relating to the radiological findings and surgery, as described above, could indirectly indicate the severity of TBI. A further study may be necessary to evaluate the influence of the severity of TBI on swallowing function.
In conclusion, the swallowing characteristics of dysphagic patients after TBI were comparable with those of dysphagic stroke patients. Common VFSS findings in patients with TBI comprised aspiration or penetration, decreased laryngeal elevation, and reduced epiglottis inversion. These results suggest that therapeutic approaches for dysphagia in stroke patients may also be applied to TBI patients with dysphagia. Patients who underwent surgical intervention after TBI tended to feed non-orally; therefore, more careful care and long-term VFSS follow-up may be required for these patients.