- Search
| Ann Rehabil Med > Volume 36(3); 2012 > Article |
Abstract
Objective
To investigate whether patterns of swallowing difficulties were associated with the location of the brain lesion, cognitive function, and severity of stroke in stroke patients.
Method
Seventy-six patients with first-time acute stroke were included in the present investigation. Swallowing-related parameters, which were assessed videofluoroscopically, included impairment of lip closure, decreased tongue movement, amount of oral remnant, premature loss of food material, delay in oral transit time, laryngeal elevation, delay in pharyngeal triggering time, presence of penetration or aspiration, and the amount of vallecular and pyriform sinus remnants. The locations of brain lesions were classified into the frontal, parietotemporal, subcortical, medulla, pons, and cerebellum. The degree of cognitive impairment and the severity of stroke were assessed by the Mini Mental Status Examination (MMSE) and the National Institute of Health Stroke Scale (NIHSS), respectively.
Results
An insufficient laryngeal elevation, the amount of pyriform sinus, and vallecular space remnant in addition to the incidence of aspiration were correlated with medullary infarction. Other swallowing parameters were not related to lesion topology. Lip closure dysfunction, decreased tongue movement, increased oral remnant and premature loss were associated with low MMSE scores. A delayed oral transit time were associated with NIHSS scores.
Post-stroke dysphagia is a very common problem; previous studies have reported that 50% of acute stroke patients experience dysphagia.1,2 The rate of ongoing dysphagia differs depending on the method used to assess the patients. A clinical assessment done 6 months after a stroke revealed that 6-11% of patients still experienced symptoms of dysphagia,3,4 and when assessed with a videofluoroscopic swallowing study (VFSS), 28-50% of the patients were confirmed to have dysphagia.4 It is recognized that for many patients, dysphagia is transient and resolves spontaneously after stroke.3 It is known that swallowing problems following acute stroke are common, and it has been documented that dysphagia in these patients may persist, resolve and then recur, or develop later in their history.3,4 Patients with dysphagia have a high incidence of malnutrition, dehydration, aspiration pneumonia, and mortality.5 Hence it is very important to assess patient for dysphagia inthe initial stages of stroke.5
Dysphagia in stroke patients causes enormous problems such the aforementioned complications, but many aspects of the neurophysiological mechanism of dysphagia have not yet been revealed. Even though it is an established fact that cerebral cortical infarction can cause dysphagia, it is still controversial which of the two hemispheres has a more important role in swallowing, and which hemisphere that is injured is likely to cause the specific patterns of dysphagia seen in stroke patients.6 Patients with lesions in the left hemisphere usually have problems in the oral phase, however patients with lesions in the right hemisphere exhibit problems with the pharyngeal phase and in particular, with aspiration.6,7 Furthermore, dysphagia lasts longer in those who have lesions in the right hemisphere than in the left.3 On the other hand, some other studies have shown no laterality by lesion location.8 A few previous studies reported that in brain stem lesions cases, impairment of the pharyngeal stage clearly is associated with this type of lesion, with the higher potential of having decreased laryngeal elevation and pharyngeal triggering time delay,9 and impairment of upper esophageal sphincter opening occurring in lateral medullary lesion.10 Nevertheless, other studies have reported that brain stem and cerebellar lesions have no statistical significant association with the incidence of dysphagia.11-13 As well, many conflicting studies about the lesion location and pattern of dysphagia relationship exist.
There is an argument that aphasia and hemineglect may also be related to dysphagia along with the lesion location.7 It was demonstrated that hemispatial neglect is related to initial dysphagia, but aphasia is unrelated to initial dysphagia.14 Therefore, the effect of lesion location and cognitive impairment of stroke patients on the patterns of dysphagia is still not clear, and in most of the studies about dysphagia, patients with aphasia and severe cognitive impairment were excluded; so there is a possibility that the relationships between those factors and dysphagia have not been properly assessed.
The purpose of this study was to study the patterns of dysphagia in every swallowing phase with VFSS, and to determine which factors, including lesion location, cognitive function, clinical characteristics (age, gender, and severity of stroke) are associated with the patterns of dysphagiain stroke patients.
Subjects of this study were the acute and subacute stroke patients who had undergone a VFSS in the Physical Medicine & Rehabilitation Department from January 2009 to December 2010 in Korea University Anam Medical Center. VFSS was done to screen for dysphagia or when a patient was referred for evaluation of swallowing difficulty. The diagnosis of stroke was limited to the cases in which cerebral infarction or hemorrhage had been verified by computed tomography (CT) or magnetic resonance imaging (MRI). Among them, patients with bilateral stroke, other diseases which could cause dysphagia, a previous history of old stroke or dementia, missing results of CT or MRI, and lesions which could not be classified such as subarachnoid hemorrhage (SAH) were excluded from the study. Patients whose VFSS were performed more than 30 days after stroke onset and those who were unable to perform an appropriate study due to their poor cooperation were also excluded from the study. Exclusion criteria also included cases in which the duration between the K-NIHSS and VFSS evaluations, and the K-MMSE and VFSS evaluations was longer than 2 weeks. A total of 76 patients satisfied the criteria and were chosen for study, and the data about these patients were collected retrospectively from the medical records.
The medical records and test results of the patients were reviewed retrospectively and their demographic and clinical characteristics were recorded parameters, such as the patient's age, gender, brain lesion laterality (left or right), type of stroke (ischemic or hemorrhagic), duration between onset of stroke and the performance of VFSS (number of days), initial severity of stroke as recorded according to the Korean version of the National Institutes of Health Stroke Scale (K-NIHSS)15,16 and duration between K-NIHSS and VFSS evaluations were investigated. Results of the Korean version of the Mini-Mental Status Examination (K-MMSE),17-19 which assessed the patient's cognitive function, were examined and duration between K-MMSE and VFSS evaluations was checked; the results of the Korean version of the modified Barthel Index (K-MBI),20,21 which assesses the independent performance of daily living activities, were also investigated. The K-NIHSS and MMSE scores of the tests that had been performed most closely to the date of the VFSS were chosen and included in the study.
Physical Medicine and Rehabilitation department conducted the VFSS by following a partly modified Logerman's.22 Subjects were seated then the fluoroscopy was performed as the patients swallowed barium mixed with orange juice, yogurt, thick gruel and rice. The studies carried out with the orange juice and yogurt were done with 2 different volumes, 2 and 5 ml. Each of the food was mixed with undiluted liquid barium for the proper bolus observation during the fluoroscopy. And when necessary, postural changes such as chin tuck positioning, head rotation, and head tilting or swallowing maneuvers such as supraglottic swallowing and the Mendelsohn maneuver were employed.
In the oral phase, functions such as lip closure, bolus formation, mastication, tongue base contact to the pharyngeal wall, tongue thrust, and premature bolus loss along with the oral transit time of the bolus were assessed. In the pharyngeal phase, laryngeal elevation or epiglottic movement, aspiration or penetration of the respiratory tract, remnants of vallecular space and pyriform sinus after swallowing were assessed, and pharyngeal transit time of the bolus, pharyngeal triggering time until the swallowing reflex appeared were observed. Each individual finding was assessed based on the presence or absence of an abnormal finding. Less than 1.25 seconds was considered a normal transit time of the bolus in the oral phase, and when it took longer, was judged abnormal. When the food entered the respiratory tract but did not pass into the true vocal fold, it was determined to be penetration, when the food otherwise passed into the true vocal fold, it was determined to be aspiration. As for pharyngeal triggering time, determined to be the time until the swallowing reflex appearance, a time of less than 0.4 seconds was considered normal. With respect to the pharyngeal transit time, less than 1 second was considered normal. The amount of remnants in the vallecular space and pyriform sinus were assessed as minimal, moderate and large; a minimal amount was considered to be normal and moderate and large amount were considered to be abnormal. We concluded the oral phase to be problematic if there was at least one lip closure abnormality in, tongue movement, oral remnant, premature loss, or oral transit time, and the pharyngeal phase to be dysfunctional if there were more than one abnormality in laryngeal elevation, pharyngeal triggering time, vallecular space or pyriform sinus remnant, penetration, or aspiration.
After analyzing the results of the brain CT or MRIs of the patients upon their visit to the hospital, the lesions were classified into two main divisions, supratentorial and infratentorial. Lesion location classification was conducted using Steinhagen's method, and among the lesion classifications, lesions involving the upper motor neurons of the cranial nerves were excluded because it is difficult to differentiate those from pontine infarction using MRI.10 The supratentorial lesions were divided into frontal lobe, parietotemporal lobe, and subcortical lesions, and the infratentorial lesions were divided into the pontine, medullary, and cerebellar lesions. Altogether there were 6 types of lesions. Lesions located in more than one position were multi-counted.
In the 6 groups that were formed on the basis of lesion locations, in order to assess whether clinical characteristics such as lesion location, laterality (left or right), gender, age, cognitive impairment and severity of stroke were independent variables in predicting the patterns of dysphagia, while controlling other possible confounding variables, corresponding rates of the findings were calculated. In order to achieve this, we set each VFSS finding as an independent variable, and the corresponding rate of each, 95% confidence interval and equivalent p-value, which was calculated with a multivariate logistic regression analysis method. A p-value of less than 0.05 was considered statistically significant. For all statistical analyses, SPSS for Windows 15.0 was used.
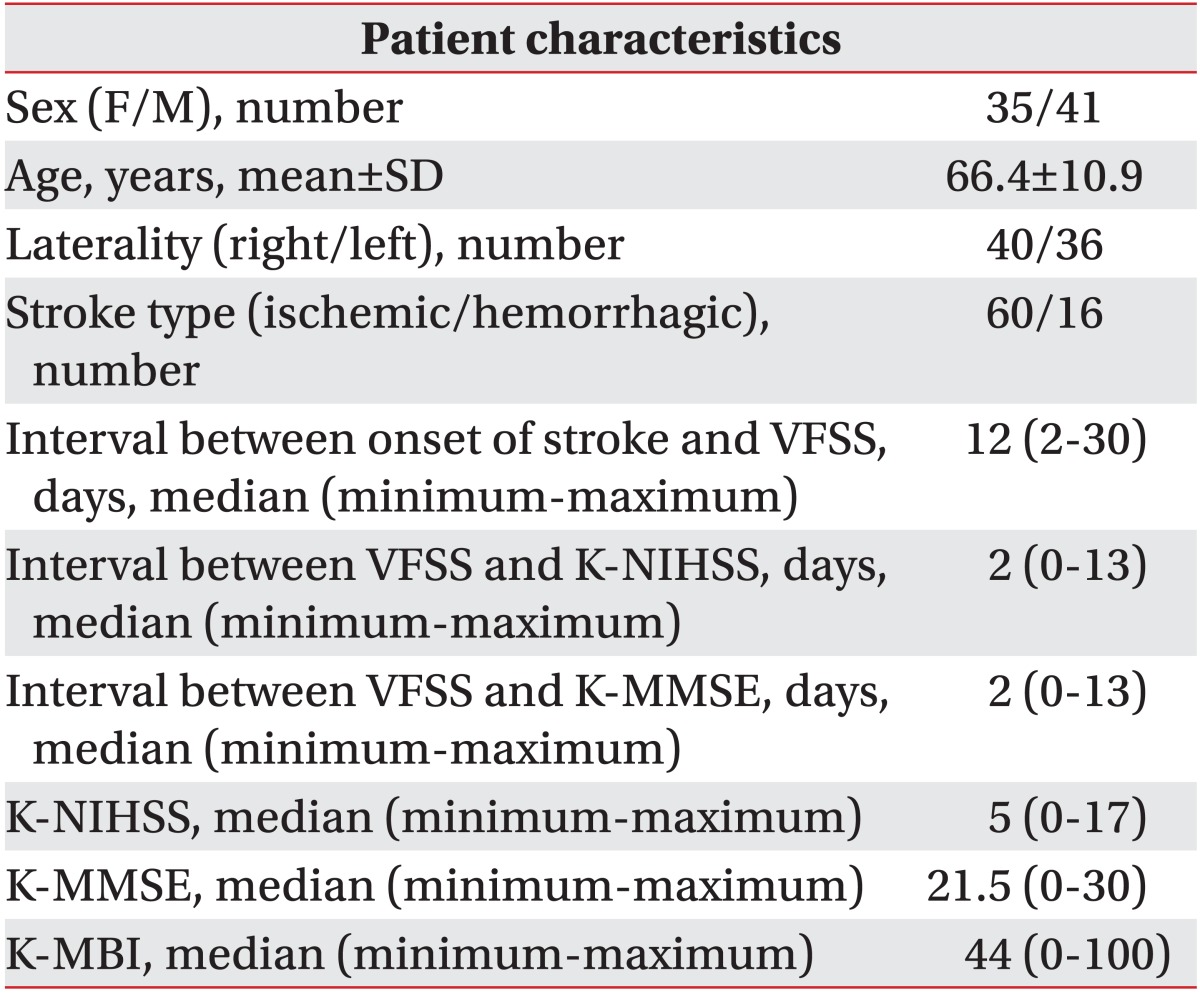
Among the 76 patients, 41 were male and 35 were female with a mean age of 66.4±10.9 years. The median duration from onset of stroke to VFSS was 12 days. The median time interval between K-NIHSS evaluation and VFSS was 2 days, and the time interval between MMSE evaluation and VFSS was 2 days.
The demographic and clinical characteristics of the 76 patients are shown in Table 1, and the characteristics of the 6 groups which had been classified by location of brain lesion are shown in Table 2.
One of the most important aspects of dysphagia is aspiration into the respiratory tract; the aspiration frequency rates in the 6 groups in descending order were as follows: medulla (45.5%), parietotempotal lobe (26.9%), frontal lobe (24.0%), pons (23.1%), cerebellum (16.6%), and subcortical lesion (13.5%) (Fig. 1). Among the groups that had been classified by lesion location, the incidence of aspiration was the highest in the medulla group (45.5%). Amongst the supratentorial lesions, aspiration incidence was highest in the parietotemporal lobe lesions.
Additionally, it was apparent that lesion laterality had no association with any of the measures of dysphagia.
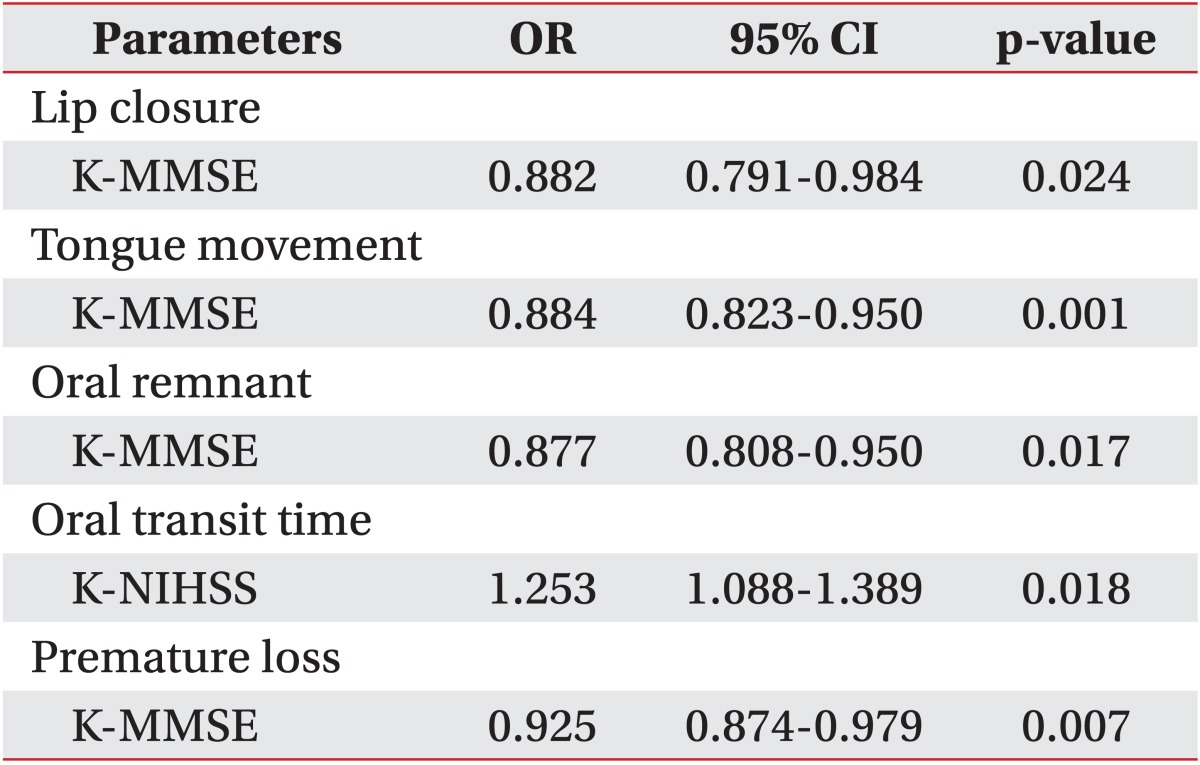
From the results of VFSS, factors relevant to dysphagia in the oral phase are described in Table 3. Dysphagia patterns in the oral phase had no relation to brain lesion location, but they were related to cognitive function and initial stroke severity. Lip closure dysfunction was related to the K-MMSE score (p=0.024). In other words, cognitive dysfunction was related to a higher incidence of lip closure dysfunction. There was a negative correlation between the incidence of tongue movement dysfunction and the K-MMSE score (p=0.001). The volume of the remnants in oral cavity was related to the K-MMSE score (p=0.017). Prolongation of oral transit time was seen more frequently as the K-NIHSS score increased (p=0.018). The factor related to premature bolus loss into the pharynx was the K-MMSE score (p=0.007).
In addition, factors which cause problems in the pharyngeal phase as observed with VFSS are described in Table 4. Problems in the pharyngeal phase were mainly related to medullary lesions. Decreased laryngeal elevation was related to two lesion areas, the medulla (p=0.000) and the parietotemporal lobe (p=0.041). It was also related to the K-MMSE (p=0.005). Therefore, on the basis of these findings, we can predict that decreased laryngeal elevation is likely to occur in patients with cognitive dysfunction and in patients who have lesions in these two areas. Aspiration was more frequent in the patients with medullary lesions (p=0.001), aspiration in these patients was seen 8 times more frequently than in the other groups. There was an increase in the frequency of remnants in the pyriform sinus after swallowing in the cases with the medullary lesion group (p=0.019). Related to frequency of remnants in the vallecular space, importantly related factors were the presence of two brain lesions, the parietotemporal lobe (p=0.047) and the medulla (p=0.046). With respect to the patient's gender, type of stroke (ischemic or hemorrhagic), and lesion laterality, we were unable to show any statistical significance to predict dysphagia patterns.
Normal swallowing involves the harmonious coordination of the oral, pharyngeal and esophageal phases. It has been argued that the location of a lesion is more important than the size or laterality of a lesion in predicting aspiration.
Studies related to dysphagia patterns in subcortical lesions showed that the occurrence of dysphagia was more frequent in left subcortical-periventricular lesions than in the right-sided ones, and a prolongation of oral transit time was the main problem.23 In a previous study, dysphagia patterns were assessed using fiberoptic endoscopic evaluations of swallowing (FEES). The study revealed that oral apraxia was present in left parietotemporal lobe lesions, impairment of the upper esophageal sphincter opening was found in lateral medullary lesions, and other swallowing measures had no relation to the location of the lesion.10
In contrast to this study, another study demonstrated that brain stem lesions showed no relation to dysphagia.11 Moreover, some other studies12,13,24 demonstrated that brain stem and cerebellar lesions had no statistical significant association with a high incidence of dysphagia.
Based on the results from the present study, we can draw a conclusion that the factors which affect the pharyngeal phase are the lesion location in the medulla and parietotemporal lobes. Similar to the findings of previous studies, our study showed that brain stem lesions, especially medullary lesions, were an independent factor associated with decreased laryngeal elevation, increased aspiration, and increased volumes of remnants in the vallecular space and pyriform sinus after swallowing. It was again confirmed that the medullary lesion location plays a positively associated role with dysphagia in the pharyngeal phase and when confounding variables (such as patient's age, gender, stroke severity, and cognitive dysfunction) are controlled.
Other than a medullary lesion, a parietotemporal lobe lesion turned out to be related to a reduced laryngeal elevation and remnants in vallecular space. Patients with lesions in the parietotemporal lobe, showed a higher percentage of stroke in the middle cerebral artery area. In many circumstances, a lesion that occupies both the primary motor and sensory cortex has either a high possibility of pharyngolaryngeal muscle hypomotility with dyskinesia or oral with pharyngeal sensory integration problems. This may demonstrate a reduced laryngeal elevation and remnants in the vallecular space for lesions in the pareitotemporal lobe cases. There also is a possibility that the involvement of corticobulbar tract, the fiber that connects the cerebral cortex and brainstem, may have caused the problem in laryngeal elevation.
None of the other dysphagia variables could be predicted on the basis of brain lesion location. In this study, most of the patients had lesions in multiple locations. As the lesion locations were multi-counted, the limitation of the present study was that the characteristics and the incidence of dysphagia between the groups could not be directly compared.
Special attention should be paid to the factors involved in the prediction of dysphagia variables in the oral phase. The problems of the oral phase occurred more frequently when the initial stroke damage was more severe and cognitive dysfunction was of a greater degree. In other words, even though there may be no aspiration during pharyngeal phase, failure of the tongue and lip to move harmoniously together or a greatly prolonged oral transit time can be an important factor for considering the insertion of a nasogastric or gastrostomy tube in a patient to ensure adequate nutrition. The factor associated with lip closure dysfunction and decreased tongue movement in the oral phase was the K-MMSE score. Also prolonged oral transit time could correlate with the K-NIHSS score. Therefore, it would be appropriate to conclude that factors causing problems with swallowing in the oral phase are cognitive dysfunction, and the initial severity of stroke. Otherwise the brain lesion location has the least relation to oral phase dysfunction.
It is well-known that in elderly patients, various physiologic changes resulting from aging and cognitive decline can bring about swallowing dysfunction. In older patients, delays in the transfer of the bolus through the oropharynx, and problems in the opening of the upper esophageal sphincter may occur.25 Esophageal motility problems can also be associated with older age, and reduced lingual pressure, reduced tongue strength, mastication problems secondary to dentition issues, slower swallowing, and increased reflux events are also related to older age.26,27 We considered that age could be potentially linked to swallowing dysfunction, but in our study, it was not a statistically significant factor for the VFSS parameters. But age itself may act as an independent variable in causing dysphagia, so special attention is needed when assessing swallowing problems in elderly patients.
In our study, we used the K-NIHSS for the evaluation of the initial severity of the stroke, but this scale was designed to assess supratentorial brain lesions, and for that reason the K-NIHSS might be a less appropriate scale for the assessment of dysphagia patients who have infratentorial lesions.28
Additionally, since some items of the K-NIHSS relate to the cognitive function of the patient,29 there is increased possibility that these patients with higher K-NIHSS scores have cognitive dysfunction. Given that the patients with high K-NIHSS tend to have dysphagia in the oral phase, cognitive function seems to be an important factor related to dysphagia. To investigate the effect of cognitive function on dysphagia, our present study, unlike most of the other studies on stroke patients, included the cases in which the K-MMSE score was remarkably low.
On the other hand, our study excluded cases in which the lesion location could not be classified according to our classification system, such as subarachnoid hemorrhage and bilateral stroke. Subsequently, there may be a chance that our study eliminated cases of serious cognitive dysfunction or serious brain damage due to SAH or bilateral stroke. Therefore, we cannot exclude the possibility that the impact of cognitive function or severity of stroke on dysphagia might have been underestimated in our study. Considering the afore-mentioned limitations along with the results from the present study, cognitive function could emerge as an important factor causing dysphagia in the oral phase.
In our study, the patient's cognitive function was assessed with the K-MMSE, a test which is simply carried out along the bedside. Due to the simplicity of the test, our present study has the limitation that a definite conclusion cannot be made about the domain of cognitive impairment that relates to dysphagia in the oral phase. In a study by Stephens et al.,30 in which the subjects' mean age was 80, the authors argued that since slight cognitive impairment cannot be entirely assessed by the MMSE, more specialized cognitive tests should be used, and poor attention has a relationship with the patient's functional prognosis. Therefore, there is a possibility that in our study the impact of cognitive function on the patterns of dysphagia might have been underestimated or distorted, because the patients with average K-MMSE scores in our study might have cognitive dysfunction in specific domains only accessible by specialized cognitive tests. In addition, another limitation of this study is the small number of subjects. When the size of subjects is small and if the subjects are analyzed by grouping, the possibility of error becomes higher.
Therefore it will be necessary to explore the relationships between specific cognitive test results and patterns of dysphagia through prospective studies carried out in the future.
Post-stroke dysphagia is a problem which causes malnutrition, dehydration and aspiration pneumonia, and it is the main reason for patients to receive nutrition via the nasogastric tube and gastrostomy. In this study, particularly in medullary lesions, pharyngeal problems such as aspiration, increase of remnants in the pyriform sinus after swallowing, and reduced laryngeal elevation were found. In brain lesions involving other areas, patterns of dysphagia cannot be predicted. The most important factor for predicting problems in the oral phase was the patient's cognitive function.
When a patient's cognitive dysfunction is severe, the incidence of oral phase dysphagia, such as reduced tongue movement, increased oral remnants, premature bolus loss, increases; therefore, dysphagia assessment is important not only for patients with a medullary lesion but also for patients who have cognitive impairment.
References
1. Daniels SK, Brailey K, Priestly DH, Herrington LR, Weisberg LA, Foundas AL. Aspiration in patients with acute stroke. Arch Phys Med Rehabil 1998;79:14-19. PMID: 9440410.


3. Smithard DG, O'Neill PA, England RE, Park CL, Wyatt R, Martin DF, Morris J. The natural history of dysphagia following a stroke. Dysphagia 1997;12:188-193. PMID: 9294937.


4. Mann G, Hankey GJ, Cameron D. Swallowing function after stroke: prognosis and prognostic factors at 6 months. Stroke 1999;30:744-748. PMID: 10187872.


5. Splaingard ML, Hutchins B, Sulton LD, Chaudhuri G. Aspiration in rehabilitation patients: videofluoroscopy vs bedside clinical assessment. Arch Phys Med Rehabil 1988;69:637-640. PMID: 3408337.

6. Robbins J, Levin RL. Swallowing after unilateral stroke of the cerebral cortex: preliminary experience. Dysphagia 1988;3:11-17. PMID: 3248391.


7. Robbins J, Levine RL, Maser A, Rosenbek JC, Kempster GB. Swallowing after unilateral stroke of the cerebral cortex. Arch Phys Med Rehabil 1993;74:1295-1300. PMID: 8259895.


8. Alberts MJ, Horner J, Gray L, Brazer SR. Aspiration after stroke: lesion analysis by brain MRI. Dysphagia 1992;7:170-173. PMID: 1499361.


9. Lee CK, Kim JA. Pattern of post-stroke swallowing disorder according to the brain lesion. J Korean Acad Rehabil Med 2001;25:193-201.
10. Steinhagen V, Grossmann A, Benecke R, Walter U. Swallowing disturbance pattern relates to brain lesion location in acute stroke patients. Stroke 2009;40:1903-1906. PMID: 19286597.


11. Falsetti P, Acciai C, Palilla R, Bosi M, Carpinteri F, Zingarelli A, Pedace C, Lenzi L. Oropharyngeal dysphagia after stroke: incidence, diagnosis, and clinical predictors in patients admitted to a neurorehabilitation unit. J Stroke Cerebrovasc Dis 2009;18:329-335. PMID: 19717014.


12. Langdon PC, Lee AH, Binns CW. Dysphagia in acute ischaemic stroke: severity, recovery and relationship to stroke subtype. J Clin Neurosci 2007;14:630-634. PMID: 17434310.


13. Broadley S, Croser D, Cottrell J, Creevy M, Teo E, Yiu D, Pathi R, Taylor J, Thompson PD. Predictors of prolonged dysphagia following acute stroke. J Clin Neurosci 2003;10:300-305. PMID: 12763332.


14. Schroeder MF, Daniels SK, McClain M, Corey DM, Foundas AL. Clinical and cognitive predictors of swallowing recovery in stroke. J Rehabil Res Dev 2006;43:301-310. PMID: 17041816.


15. Lyden PD, Lu M, Levine SR, Brott TG, Broderick J. A modified National Institutes of Health Stroke Scale for use in stroke clinical trials: preliminary reliability and validity. Stroke 2001;32:1310-1317. PMID: 11387492.


16. Lee KM, Jang YH, Kim YH, Moon SK, Park JH, Park SW, Yu HJ, Lee SG, Chun MH, Han TR. Reliability and validity of Korean Version of National Institutes of Health Stroke Scale. J Korean Acad Rehabil Med 2004;28:422-435.
17. Mungas D, Marshall SC, Weldon M, Haan M, Reed BR. Age and education correction of Mini-Mental State Examination for English and Spanish-speaking elderly. Neurology 1996;46:700-706. PMID: 8618670.


18. Kohn R, Vicente B, Rioseco P, Saldivia S, Torres S. The mini-mental state examination: age and education distribution for a Latin American population. Aging Ment Health 2008;12:66-71. PMID: 18297480.


19. Kim SJ, Park YH. Comparison of sensitivity of neurobehavioral cognitive status examination and minimental state examination. J Korean Acad Rehabil Med 1997;21:1105-1109.
20. Granger CV, Dewis LS, Peters NC, Sherwood CC, Barrett JE. Stroke rehabilitation: analysis of repeated Barthel index measures. Arch Phys Med Rehabil 1979;60:14-17. PMID: 420565.

21. Jung HY, Park BK, Shin HS, Kang YK, Pyun SB, Paik NJ, Kim SH, Kim TH, Han TR. Development of the Korean Version of Modified Barthel Index (K-MBI): multicenter study for subjects with stroke. J Korean Acad Rehabil Med 2007;31:283-297.
22. Logemann JA. Swallowing physiology and pathophysiology. Otolaryngol Clin North Am 1988;21:613-623. PMID: 3054716.


23. Cola MG, Daniels SK, Corey DM, Lemen LC, Romero M, Foundas AL. Relevance of subcortical stroke in dysphagia. Stroke 2010;41:482-486. PMID: 20093638.


24. Paciaroni M, Mazzotta G, Corea F, Caso V, Venti M, Milia P, Silvestrelli G, Palmerini F, Parnetti L, Gallai V. Dysphagia following Stroke. Eur Neurol 2004;51:162-167. PMID: 15073441.


26. Daggett A, Logemann J, Rademaker A, Pauloski B. Laryngeal penetration during deglutition in normal subjects of various ages. Dysphagia 2006;21:270-274. PMID: 17216388.


27. Leslie P, Drinnan MJ, Ford GA, Wilson JA. Swallow respiratory patterns and aging: presbyphagia or dysphagia? J Gerontol A Biol Sci Med Sci 2005;60:391-395. PMID: 15860480.



28. Sato S, Toyoda K, Uehara T, Toratani N, Yokota C, Moriwaki H, Naritomi H, Minematsu K. Baseline NIH Stroke Scale Score predicting outcome in anterior and posterior circulation strokes. Neurology 2008;70:2371-2377. PMID: 18434640.


29. Cumming TB, Blomstrand C, Bernhardt J, Linden T. The NIH stroke scale can establish cognitive function after stroke. Cerebrovasc Dis 2010;30:7-14. PMID: 20424439.


30. Stephens S, Kenny RA, Rowan E, Kalaria RN, Bradbury M, Pearce R, Wesnes K, Ballard CG. Association between mild vascular cognitive impairment and impaired activities of daily living in older stroke survivors without dementia. J Am Geriatr Soc 2005;53:103-107. PMID: 15667385.


Fig. 1
Frequency of aspiration according to the brain lesion. The aspiration frequency rates in the 6 groups in a descending order were as follows: medulla (45.5%), parietotempotal lobe (26.9%), frontal lobe (24.0%), pons (23.1%), cerebellum (16.6%), and subcortical lesion (13.5%). P-T: Parietotemporal.
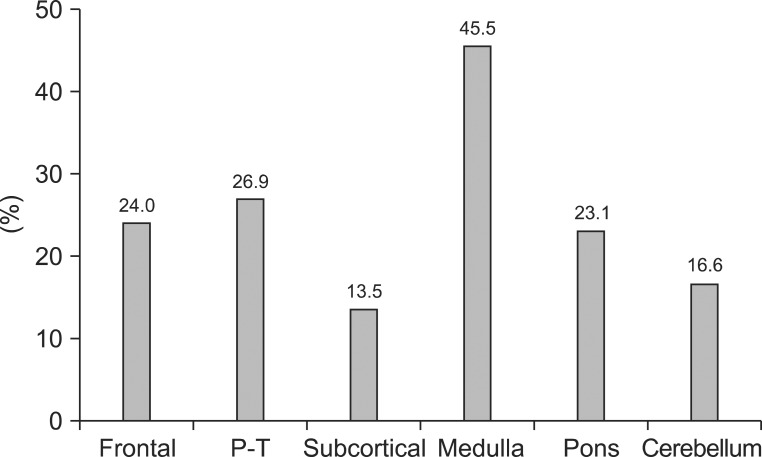
Table 2
Demographic and Clinical Characteristics of 6 Subgroups (n=76)
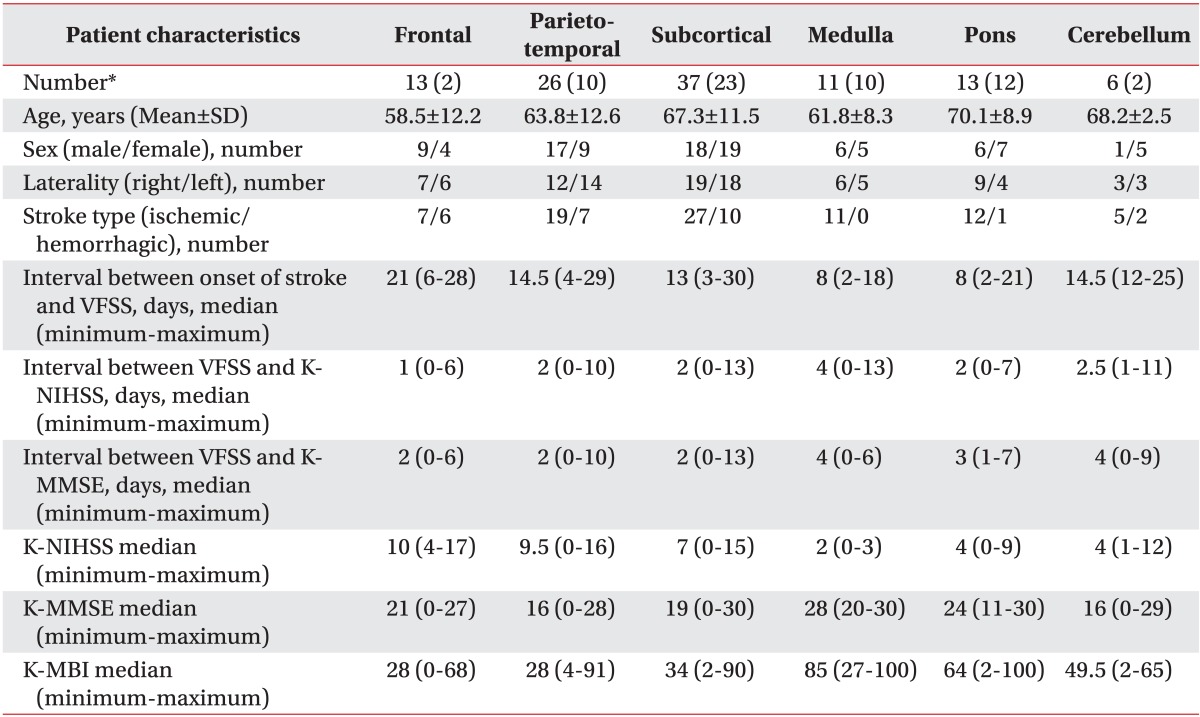
K-NIHSS: Korean version of National Institute of Health Stroke Scale, VFSS: Videofluoroscopic swallowing study, K-MMSE: Korean version of Mini Mental Status Examination, K-MBI: Korean version of modified Barthel Index
*Number of patients with combined (isolated) infarction in the referring brain lesion